☐ | Preliminary Proxy Statement |
☐ |
|
☒ | Definitive Proxy Statement |
☐ | Definitive Additional Materials |
☐ | Soliciting Material Pursuant to Section 240.14a-12 |


☒ | No fee required. |
☐ | Fee paid previously with preliminary materials. |
☐ | Fee computed on table in exhibit required by Item 25(b) per Exchange Act Rules 14a-6(i)(1) and0-11 |




| ÷ ÷ ÷ ÷ | 1 |
Notice of Annual Meeting
of Shareholders
To Merck Shareholders:
You are invited to the Annual Meeting of Shareholders of Merck & Co., Inc. (the “Company” or “Merck”) on Tuesday, May 28, 2024, at 9:00 a.m. (Eastern Time) via Webcast at www.virtualshareholdermeeting.com/MRK2024 (the “2024 Annual Meeting”).
The purposes of the meeting are to:
1. Elect the 12 Director nominees named in this proxy statement;
2. Consider and act upon a proposal to approve, by non-binding advisory vote, the compensation of our Named Executive Officers;
3. Consider and act upon a proposal to ratify the appointment of PricewaterhouseCoopers LLP as the Company’s independent registered public accounting firm for 2024;
4. Consider and act upon a shareholder proposal regarding a shareholder right to act by written consent, if properly presented at the meeting;
5. Consider and act upon a shareholder proposal regarding a government censorship transparency report, if properly presented at the meeting;
6. Consider and act upon a shareholder proposal regarding a report on respecting workforce civil liberties, if properly presented at the meeting; and
7. Transact such other business as may properly come before the meeting.
By order of the Board of Directors,
Kelly E. W. Grez
Corporate Secretary
| ||||
| ||||
| ||||
Vote Right Away—Advance voting methods and deadlines
We encourage all shareholders of record to read this proxy statement with care and vote right away using any of the following methods, even if they intend to attend the 2024 Annual Meeting. In all cases, have your proxy card or voting instruction form in hand and follow the instructions.
| ||||||||
 | BY INTERNET* | www.proxyvote.com | ||||||
 | BY PHONE* | In the U.S. or Canada dial toll-free 1-800-690-6903 | ||||||
 | BY QR CODE | |||||||
 |
to be directed to www.proxyvote.com | |||||||
 | BY MAIL** | Cast your ballot, sign your proxy card and send in our prepaid envelope | ||||||
Only shareholders listed on the Company’s records at the close of business on
Merck began distributing its Notice of Internet Availability of Proxy Materials, proxy statement, the
IMPORTANT NOTICE REGARDING THE AVAILABILITY OF PROXY MATERIALS FOR THE ANNUAL MEETING OF SHAREHOLDERS TO BE HELD ON
The Notice of Annual Meeting of Shareholders, proxy statement and the The principal executive offices of the Company are located at * The telephone and internet voting facilities will close at 11:59 p.m. Eastern Time on May 27, 2024. ** If your shares are held in a stock brokerage account or by a bank or other nominee, your ability to vote by telephone or over the internet depends on your broker’s voting process. Please follow the directions provided to you by your broker, bank or nominee.
| ||||||||
* The telephone and internet voting facilities will close at 11:59 p.m. Eastern Time on May 23, 2022.
** You will need the 16-digit control number included on your proxy card, voting instruction form or Notice of Internet Availability of Proxy Materials.
If your shares are held in a stock brokerage account or by a bank or other nominee, your ability to vote by telephone or over the internet depends on your broker’s voting process. Please follow the directions provided to you by your broker, bank or nominee.
*** Effective May 1, 2022, the Company’s headquarters will be relocated to Rahway, N.J. and the principal executive offices of the Company will be located at 126 East Lincoln Avenue, Rahway, N.J. 07065 U.S.A.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 2 | ||||||||||
ç ç ç ç |
|
Dear Merck Shareholders,
It is2023 was a very strong year for our pleasurecompany as we continued to invitedeliver for all our stakeholders, including patients, society and you, to the 2022 Annual Meetingour shareholders. Driven by our purpose of Shareholders of Merck & Co., Inc. (“Merck,” known as “MSD” outside the United States and Canada).
The foregoing Notice of Annual Meeting of Shareholders and accompanying proxy statement will serve as your guide to the business to be conducted and provide details regarding the meeting.

For more than 130 years, Merck has usedusing the power of leading-edge science to deliver products that save and improve lives. We remain committedlives around the world, our team worked with urgency, rigor and passion to this purpose asdevelop and deliver important scientific advancements. I am confident our science-led strategy, which keeps the patient at the center of everything we do, will enable us to continue to positively impact global healthdriving value creation today and for generations to come. Aswell into the COVID-19 pandemic continued throughout 2021, we prioritized protectingnext decade.
Over the safety of our employees and their families, sustaining the supply of our medicines and vaccines, and supporting patients in our clinical trials. Our inspiration, innovation and ingenuity enabled us to play a leading role in bringing forward important medicines and vaccines to address many of the world’s most challenging diseases, including our investigational antiviral drug molnupiravir, adding to the short list of important medicines that are part of the global effort in the fight against COVID-19.
Throughout last year, we continued to transformadvance our business, driving greater focus on our innovative portfoliopriority programs and increasing our operational efficiency. The successful spin-offtook meaningful steps to build one of Organon is just one notable example. Today, as a result of our strategicthe broadest and dynamic actions, our company is more focused, more efficient and faster-growing. Guided by our business and capital allocation strategy, we continued to invest in the discovery, development, production and commercialization of medicines and vaccines. This strengthened the short- and long-term sustainability of our business, and accelerated and augmented our pipeline and portfolio.
From a commercial standpoint, our teams executed at the highest levels, achieving strong growth across our key performance drivers, including KEYTRUDA – an established and foundational assetmost diversified pipelines in our portfolio –recent history. We initiated more than 20 Phase 3 studies, which included advancing eight novel candidates. In 2024, we anticipate an even greater number of Phase 3 study starts and multiple data readouts. We also look forward to three potential new product approvals with the promise to provide significant patient benefit.
In oncology, September 2024 marks a decade since the first approval of KEYTRUDA, a foundational medicine for the treatment of certain types of cancer, currently approved by the U.S. Food and Drug Administration (FDA) for 39 indications across 17 different tumor types. We are committed to continuing to provide important innovation to patients and maintaining our leadership in oncology. Our strong, diverse oncology pipeline includes candidates spanning immuno-oncology, precision molecular targeting and tissue-targeting agents.
As we continue to realize the potential of KEYTRUDA, we are focusing increasingly on earlier stages of disease, where effective intervention has the potential to improve outcomes. We made progress in 2023 for the treatment of certain patients with non-small cell lung cancer (NSCLC) with FDA approvals for KEYNOTE-091 as well as KEYNOTE-671, which met its dual primary endpoints of event-free survival and overall survival. In January 2024, we announced FDA approval for KEYTRUDA in vaccinescombination with GARDASIL
“I am confident our science-led strategy, which keeps the patient at the center of everything we do, will enable us to continue driving value creation today and GARDASIL 9,well into the next decade.”
chemoradiotherapy for the treatment of stage III through IVA high-risk, locally advanced cervical cancer, based on the Phase 3 KEYNOTE A-18 trial. In the earlier-stage setting, we and our partner Moderna announced positive three-year recurrent-free survival and distant metastasis-free survival data for our individualized neoantigen therapy, V940, in Animal Health. Overall, it has beencombination with KEYTRUDA for the adjuvant treatment of certain patients with stage III and IV melanoma following complete resection. Together with collaborators, we also announced FDA approval for KEYTRUDA in combination with Padcev, a year of significant achievementtreatment for Merck in the faceadult patients with locally advanced or metastatic urothelial cancer.
We’re making progress with our precision molecular targeting and tissue targeting oncology molecules. The FDA approval of an extraordinarily challenging environment.
Weadditional indication for WELIREG, our HIF-2a inhibitor for the treatment of adults with advanced our oncology portfoliorenal cell carcinoma following a PD-1 or PD-L1 inhibitor and made substantiala VEGF-TKI, marks the first approval in a novel therapeutic class for this population in nearly a decade. In addition, we showed meaningful progress in executing our strategy to become the leadingrobust oncology company by 2025.pipeline, initiating Phase 3 trials for four investigational medicines, including bomedemstat (LSD1 inhibitor), nemtabrutinib (BTK inhibitor), MK-2870 (anti-TROP2 antibody-drug conjugate, or ADC) and MK-5684 (CYP11A1 inhibitor). We were pleased to receive FDA approvals in several women’s cancers,have built a pipeline of ADCs through our collaborations with Kelun-Biotech and Daiichi Sankyo as well as renal cell carcinoma (adjuvantour own discovery programs.
Our growing cardiometabolic pipeline is another exciting area of focus, one where we see significant long-term potential. We recently received FDA approval for WINREVAIR (sotatercept-csrk), a first-in-class activin signaling inhibitor biologic for the treatment of adults with pulmonary arterial hypertension (PAH). PAH is a rare, progressive and advanced)potentially devastating disease of the blood vessels in the lungs, which has a profound impact on patients’ lives and melanoma (adjuvant) for KEYTRUDA. Weon their life expectancy. Additionally, we are excited that Merck has more than 90 potential approvals in oncology on the horizon. In 2022, our work in women’s and earlier stage therapies continues with additional focus on advancing prostate and colorectal cancer treatments.
|
We are proud that upon receiving Emergency Use Authorization from the FDA in late December 2021, we successfully delivered initial molnupiravir shipments to the U.S. government and to other countries where molnupiravir has been authorized for use, including the UK and Japan. This is a testament to Merck’s agility and ability to establish critical partnerships with governments, public health agencies, key global stakeholders, and across various links in the international supply chain. Our ground-breaking access strategy has been a priority from the start and has accelerated the timely distribution of molnupiravir to patients in need globally.the European Union, where we expect regulatory action in the second half of 2024.
Beyond WINREVAIR, promising candidates like MK-0616, a novel oral PCSK9 inhibitor for the treatment of hypercholesterolemia currently in Phase 3 trials, and MK-6024, an investigational GLP-1/glucagon receptor co-antagonist being evaluated for the treatment of metabolic dysfunction-associated steatohepatitis (MASH), currently in Phase 2 trials, reinforce our expectation that we can achieve positive impact for patients and the potential for approximately $15 billion in revenue from medicines to treat cardiometabolic disease by the mid-2030s.
In our vaccines business, worldwide demand for our vaccines for the prevention of certain human papillomavirus (HPV)-related cervical cancers and other diseases – GARDASIL
Merck & Co., Inc. 2022 2024 Proxy Statement
| ÷ ÷ ÷ ÷ | 3 |
In line with our access strategy, we allocated up to 3 million courses of molnupiravir to UNICEF throughout the first half of 2022 for distribution in more than 100 low- and middle-income countries. This was in addition to our agreements with the Medicines Patent Pool and other generic manufacturers to make molnupiravir available following local regulatory authorizations or approvals.
Our Animal Health business continues to expand with growth across species and geographies. Growth in companion animal product sales were led by the BRAVECTO parasiticide line and our line of companion vaccines, while livestock animal product sales showed higher demand globally in the poultry, ruminant and swine sectors. The Animal Health business remains very well positioned to grow faster than the overall market well into the future.
We are confident in the strength and durability of Merck’s business model for this decade and beyond, and we are laser-focused on executing to achieve our goals for long-term growth and sustainability. We have important growth drivers with our durable products that include GARDASIL and GARDASIL 9 which we believe can double sales by 2030. In addition– continues to our HPV vaccines, our vaccines portfolio and pipeline are growing, including with VAXNEUVANCE, which is under priority review by the FDA to expand its use to infants and children for the prevention of invasive pneumococcal disease.
We further strengthened our late-stage pipeline through our acquisition of Acceleron Pharma, bringing in sotatercept which addresses pulmonary arterial hypertension, as well as other compounds designed to address areas of serious unmet need in cardiovascular disease. These new additions complement our growing internal cardiometabolic pipeline of new drugs. In addition to Acceleron, we also completed the acquisition of Pandion Therapeutics, which enabled us to bring in an early-stage asset, leverage our immunology learnings from our immuno-oncology research and begin to extend our focus into autoimmune diseases. Business development will remain an imperative strategic priority that helps bolster and augment our pipeline, drives stronger performance, and enhances our long-term potential. We will continue to aggressively pursue compelling external science to supplement our internal pipeline, leveraging a science-driven and portfolio-informed approach.
|
Merck will continue improving patient outcomes and contributing to a healthier world, while also delivering value for our shareholders and all stakeholders.grow. We are proud of our legacy and leadership in the prevention of certain HPV-related cancers and diseases, and we expect to continue to protect millions more people around the world. Also in vaccines, the FDA granted Priority Review for our biologics license application for V116. If approved, V116 would be the first vaccine specifically designed to address the serotypes responsible for approximately 83% of invasive pneumococcal disease in adults ages 65 and older. We anticipate launching V116 in the second half of 2024, and we are confident that V116 represents a multibillion-dollar opportunity.
Our Animal Health business achieved solid growth in 2023, driven by balanced performance across both livestock and companion animal products. Our recently announced agreement to acquire the aqua business of Elanco Animal Health will, upon closing, establish Merck Animal Health as a leader in this business segment. We are well positioned in Animal Health with a strong pipeline of livestock and companion animal products, and we expect to achieve above-market growth over the long term.
Business development remains a priority and an integral element of our science-led business strategy at Merck, and in 2023, we built on our strong track record of identifying and accessing the best science to enhance our pipeline and drive long-term growth with several strategic acquisitions and new collaborations. We accelerated our presence in immunology with the acquisition of Prometheus Biosciences, and subsequently initiated a Phase 3 study for tulisokibart (MK-7240), a TNF-like ligand 1A (TL1A) antibody, in patients with ulcerative colitis. We announced a collaboration with Daiichi Sankyo for three potential first-in-class ADCs that provide the opportunity to develop meaningful new options for patients with certain types of cancer and the opportunity to deliver the next generation of precision cancer medicines. We are also excited about our acquisition of Imago BioSciences, which expands our hematology presence. In addition, our 2024 acquisition of Harpoon Therapeutics augmented our oncology pipeline with a novel portfolio of T-cell engagers, including lead
candidate MK-6070, a T-cell engager targeting delta-like ligand 3 (DLL3), which is being evaluated in certain types of small-cell lung cancer and neuroendocrine tumors.
We remain deeply passionate about the work we do to enable access and have made strategic commitments to ensure a positive impact on global health. In 2022, we surpassed our future,goal to enable 100 million more people to access our innovative portfolio of medicines and vaccines – three years ahead of schedule. As a result, we have raised our ambition and set a new goal to enable access for 350 million people. We have implemented several critical strategies to realize this goal, one of which focuses on collaboration with key financial institutions and payers, helping them expand funding options that assist patients and their families with managing out-of-pocket medical costs due to critical illness. Additionally, guided by our purpose and principles, we continue to uphold our long-term commitment of pricing our medicines responsibly.
Internally and across our global enterprise, we consistently invest in our colleagues, foster a positive and inclusive working environment and improve representation across all dimensions of diversity. Importantly, in 2022 women represented over half of our new hires globally, and 47% of new hires in the U.S. came from underrepresented ethnic groups. In the U.S., we also achieved greater than 99% pay equity for female and male employees, as well as non-white (including Black, Hispanic and Asian employees) and white employees.
In 2024, we work with speed, urgency,will continue to leverage our size, scope and agilityscale to bring forward innovations that address unmet needs. For over a century, Merck has been propelled by bold ideasgrow and innovation that advance human health. We know the world needs more of what Merck can deliver now –our pipeline; and capitalize on opportunities to provide lifesaving and life-changing medicines and vaccines to patients worldwide. Ultimately, this is what inspireswill enable us to continue helpingdeliver value to patients, employees, health care professionals, shareholders and healing patients aroundall our stakeholders. On behalf of the world as we move forward.
ThankCompany and the Board, thank you for your confidence andongoing support of our Company.company. We hopeappreciate your partnership and perspectives and encourage you willto participate in the Annual Meeting by attending virtually or by voting, as promptly as possible, through other acceptable means as described in this proxy statement. Your participation is important, so please exerciseexercising your right to vote.
 |
|  |
Robert M. Davis Chairman, Chief Executive Officer and President | “ In 2024, we will continue to leverage our size, and vaccines to patients worldwide.” |
Merck & Co., Inc. 2022 2024 Proxy Statement
| 4 | ||||||||||
ç ç ç ç |
|
A Message from Merck’s Independent Lead Director
Dear Merck Shareholders,
For more than 130 years, MerckAs our Chairman and CEO, Robert M. Davis, shared in his letter, 2023 was a very strong year for the Company as it continued working to fulfill its purpose of using the power of leading-edge science to save and improve lives around the world. This purpose has remained dedicated toguided the Company throughout its mission of saving and improving lives. This dedication is evident in Merck’s commitment to providing timely access globally, following applicable authorizations and approvals, to molnupiravir,long history. It also guides the Company’s investigational oral antiviral COVID-19 medicine. Merck has pursued a comprehensive supply and access approach to fulfilling this commitment, including investing at risk to produce millions of courses of therapy and granting a voluntary license to the Medicines Patent Pool to make generic molnupiravir available in more than 100 low- and middle-income countries following local regulatory authorizations or approvals – the first such license to the Medicines Patent Pool for a COVID-19 medical technology.
My fellow Directors and I are committed to Merck’s mission as wellBoard in our work overseeing the Company’s affairs and fulfilling our responsibilities. The process of planning and executing a smooth CEO transition, in particular, is one of the Board’s most important responsibilities. Our planning resulted last year in a successful CEO transition with Robert M. Davis, then-CFO, succeeding Kenneth C. Frazier as the Company’s President, effective April 1, 2021, and Chief Executive Officer, effective July 1, 2021. Mr. Davis also became a Board member effective July 1, 2021. The Board believes Mr. Davis is the right person to lead Merck into the future and continue its long history of focusing on science and innovation as the driver of long-term sustainable value creation for patients and shareholders. In September 2021, I was honored to be selected by my fellow independent Directors to assume the role of Lead Director. We also announced other important leadership transitions in 2021, electing Dean Y. Li to succeed Roger M. Perlmutter as Executive Vice President and President, Merck Research Laboratories, and Caroline Litchfield to succeed Mr. Davis as Chief Financial Officer. Most recently, with the Board’s support, the Company announced a new leadership structure for its Human Health business, consisting of (i) Human Health Global Marketing led by Arpa Garay, (ii) Human Health U.S. led by Jannie Oosthuizen, and (iii) Human Health International led by Deepak Khanna on an interim basis. This structure will enable the Company to build on its momentum, develop its internal talent and expand its high performing senior leadership team.
In addition to management succession planning, the independent members of the Board regularly review the Board’s leadership structure and will do so again in 2022. Our Board believes that our shareholders and our Company are best served by allowing the Board to exercise its judgment regarding the most appropriate leadership structure at a given time. As part of the Company’s CEO transition, and considering the facts and circumstances at the time, the Board determined that Merck’s shareholders were best served by a leadership structure consisting of (i) Mr. Frazier, our former CEO, serving as Executive Chairman for a transition period, (ii) Mr. Davis serving as CEO and President, and (iii) an independent director appointed by the Board’s independent members serving as Lead Director, and each role has clearly delineated responsibilities. For example, as Executive Chairman, Mr. Frazier focuses on Board operations and governance matters, as CEO, Mr. Davis manages the general supervision, direction and strategy of the business and affairs of the Company subject to the Board’s overall oversight, and, as independent Lead Director, I work closely with both our Executive Chairman and our CEO to set board agendas, approve board materials and ensure that Merck achieves the highest level of corporate governance.
Both as a full Board and through our four standingindependent committees, composed of independent directors only, we are dedicated to the effective oversight of the Company’s strategy and business operations and the key risks it faces. Board leadership is an important component of ensuring effective oversight. We review the Board’s leadership structure at least annually to confirm the current structure remains the most appropriate leadership structure for the Company faces. Weand the Board at a given time. The Board continues to believe inthat having Mr. Davis serve as Chairman and CEO provides strategic and operational expertise and perspective to the business valueChairman role because he can draw on his detailed institutional knowledge of the Company and his industry experience. At the same time, we have strong independent oversight through (a) the key duties and responsibilities I discharge as independent Lead Director and (b) our four independent Board committees chaired by independent Directors. Together, we ensure that Merck achieves the highest level of corporate governance, which includes having diverse perspectives in the boardroom. We are deliberate in ensuring we haveboardroom and dialogue with shareholders.
Board composition is, of course, essential to effective oversight. My fellow Directors and I believe that our twelve Director nominees possess broad expertise, skills, experience, and perspectives that will facilitate the right mix of perspectives, skillsstrong oversight and expertisestrategic direction required to addressgovern the Company’s currentbusiness and anticipated needs as opportunitiesstrengthen and challenges facingsupport senior management. To help demonstrate this, and in response to shareholder feedback, this proxy statement includes not only an individualized skills matrix showing the Company evolve. Our Directorskey skills that each Director represents, but also a matrix sharing demographic information for each Director. In fulfilling our board responsibilities, we draw on theirour unique experiences to provide guidance on corporate strategy and monitor its implementation in areas such as research and development, capital allocation, risk management, operating results, human capital management and global manufacturing. The
In addition, our annual self-evaluation process helps us identify ways to continue to enhance the overall effectiveness of the Board and our committees. It also provides oversight forgives us the Company’s ESG strategy and performance as a whole andopportunity to discuss other important topics through feedback from each of our committees based on their specific areas of competency. This year, we are delighted to nominate as a new Director, Douglas M. Baker, Jr., Executive Chairman of Ecolab, Inc., a provider of water and hygiene services and technologies forDirectors that is then considered by the food, hospitality, industrial, and energy markets. Mr. Baker brings extensive expertise in corporate governance and general and organizational management, and we look forward to him joining thefull Board.
We appreciate your investment in Merck and your support for the Board. We remain committed to serving you and the patients around the world that depend on Merck’s life-saving work.
  |
Thomas H. Glocer Independent Lead Director April 11, 2024 |
Merck & Co., Inc. 2022 2024 Proxy Statement
| ÷ ÷ ÷ ÷ | 5 |
| Proxy Summary | 6 | ||||||
| Corporate Governance | 11 | ||||||
11 | |||||||
12 | |||||||
13 | |||||||
14 | |||||||
17 | |||||||
18 | |||||||
| |||||||
| |||||||
| |||||||
Board Succession Planning, Criteria for Board Membership, and Director Nomination Process | 20 | ||||||
| 22 | ||||||
23 | |||||||
| |||||||
| |||||||
24 | |||||||
| 25 | |||||||
Access and Pricing Transparency for Our Medicines and Vaccines |
| 26 | |||||
Political Contributions and Lobbying Expenditure Oversight and Disclosure |
| ||||||
| 30 |
| ||||||
| |||||||
| |||||||
| |||||||
| Stock Ownership Information | |||||||
| |||||||
| |||||||
| |||||||
| Proposal 1. Election of Directors | |||||||
| |||||||
| Director Compensation | 42 | ||||||
| |||||||
| Proposal 2. Non-Binding Advisory Vote to Approve the Compensation of Our Named Executive Officers | |||||||
| Compensation Discussion and Analysis | |||||||
| |||||||
Executive Compensation |
| ||||||
| 47 |
| ||||||
| |||||||
| |||||||
| |||||||
| |||||||
| |||||||
Merck & Co., Inc. 2022 2024 Proxy Statement
|
6 |
| ||||||||||
| | | | |
| |||||||||||
This summary highlights information contained elsewhere in this proxy statement and does not contain all of the information that you should consider. You should read the entire proxy statement carefully before voting.
Date and Time Tuesday, May 9:00 a.m. ET | Record Date
April 1, 2024 | |
Location Via Webcast at www.virtualshareholdermeeting.com/
| ||
Voting Matters | Board’s Recommendation | |||||
Proposal 1 Election of Directors | FOR each Nominee | |||||
Proposal 2 Non-binding Advisory Vote to Approve the Compensation of our Named Executive Officers (Say-on-Pay) | FOR | |||||
Proposal 3 Ratification of Appointment of Independent Registered Public Accounting Firm for | FOR | |||||
Shareholder Proposals |
|
| ||||
Shareholder | AGAINST | |||||
| ||||||
| ||||||
Business Highlights
|


| $ | in total R&D expenses in
|
| |||
Capital Returned & Dividend Increase
|
$ 8.8B | Capital Returned to Shareholders (dividends and share repurchases)
|
| |||

Annualized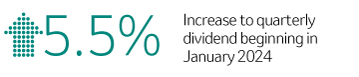
Total Shareholder Return(1)
Year-End 2021 | ||
| 1-Year | 3-Year | |
| 1.8% | 4.9% | |
| 5-Year | ||
9.8%
| ||
(1) Relative Total Shareholder Return, a component of our Performance Share Unit program that is described on page 55, is calculated on a different basis.
| ||
Year-End 2023 | ||
| 1-Year | 3-Year | |
| 1.0% | 15.4% | |
| 5-Year | ||
11.7%
| ||
(1) Relative Total Shareholder Return, a component of our Performance Share Unit program that is described on pages 56-57, is calculated on a different basis.
| ||
Merck & Co., Inc. 2022 2024 Proxy Statement
Proxy Summary | ÷ ÷ ÷ ÷ | 7 |
20212023 NEOs and Compensation Highlights (Page 48)50)
Below is a list of our 20212023 Named Executive Officers, or “NEOs”, and select compensation highlights from 2021.2023. For additional information on our elements of 20212023 compensation, please refer to the Compensation Discussion and Analysis (“CD&A”), beginning on page 43.45.
Annual Base Salary$
|
Target Annual Incentive%
|
Target Long-Term
|
Target TDC Increase%(1)
| |||||||||||||||||||||
|
2021 NEOs
|
| ||||||||||||||||||||||
|
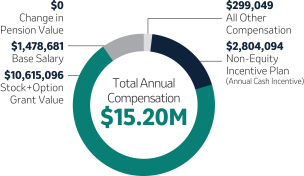
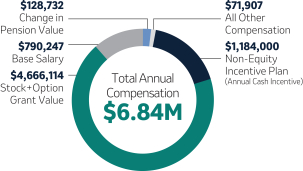
Robert M. Davis Chief Executive Officer, President and Former Chief Financial Officer | $1,500,000 | 150% | $10,750,000(2) | +131% | |||||||||||||||||||
......................................................................................................................................................... | ||||||||||||||||||||||||
 |
Kenneth C. Frazier Executive Chairman and Former Chief Executive Officer
| 1,250,000 | 100 | 10,750,000 | -34 | |||||||||||||||||||
......................................................................................................................................................... | ||||||||||||||||||||||||
 |
Caroline Litchfield Executive Vice President and Chief Financial Officer
| 900,000 | 100 | 2,200,000 | —(3) | |||||||||||||||||||
......................................................................................................................................................... | ||||||||||||||||||||||||
 |
Frank Clyburn Former Executive Vice President and President, Human Health(4)
| 1,000,000 | 100 | 4,300,000 | —(3) | |||||||||||||||||||
......................................................................................................................................................... | ||||||||||||||||||||||||
 |
Richard R. DeLuca, Jr. Executive Vice President and President, Merck Animal Health
| 800,000 | 100 | 2,700,000 | —(3) | |||||||||||||||||||
......................................................................................................................................................... | ||||||||||||||||||||||||
 |
Dean Li, M.D., Ph.D. Executive Vice President and President, Merck Research Laboratories
| 950,000 | 100 | 3,000,000 | —(3) | |||||||||||||||||||
Annual Base Salary$(1)
|
|
Target Annual Incentive%
|
|
Target Long-Term
|
|
Target TDC Increase%(2)
| ||||||||||||||||||
|
2023 NEOs |
| ||||||||||||||||||||||
|
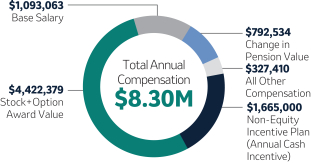
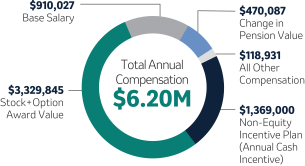
Robert M. Davis Chairman, Chief Executive Officer and President | $1,615,000 | 150% | $13,500,000 | +12.3% | |||||||||||||||||||
............................................................................................................................................................................. | ||||||||||||||||||||||||
 |
Caroline Litchfield Executive Vice President and Chief Financial Officer
| 1,125,000 | 100 | 4,250,000 | +38.3 | |||||||||||||||||||
............................................................................................................................................................................. | ||||||||||||||||||||||||
 | Sanat Chattopadhyay Executive Vice President and President, Merck Manufacturing Division
| 941,806 | 100 | 3,300,000 | —(3) | |||||||||||||||||||
............................................................................................................................................................................. | ||||||||||||||||||||||||
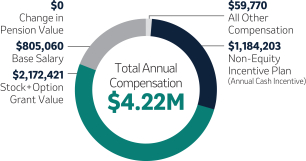
 | Richard R. DeLuca, Jr. Executive Vice President and President, Merck Animal Health
| 925,000 | 100 | 3,200,000 | +7.4(3) | |||||||||||||||||||
............................................................................................................................................................................. | ||||||||||||||||||||||||
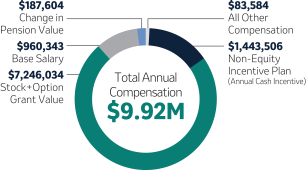
 | Dean Li, M.D., Ph.D. Executive Vice President and President, Merck Research Laboratories
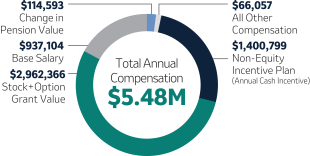
| 1,400,000 | 100 | 5,600,000 | +31.3 | |||||||||||||||||||
| (1) | Reflects base salary as of December 31, 2023. |
| (2) | Target Total Direct Compensation (“TDC”) is defined as the sum of annual base salary, target annual cash incentive and target long-term incentive. This column reflects the increase in Target TDC from |
|
| (3) |
|
|
Variable Compensation is Critical to Achieve Our Objectives (Page 45)53)
Merck’s compensation programs are designed to align the interests of our executives with the interests of our shareholders, among other objectives. For this reason, a significant portion of our NEOs’ pay is variable and at-risk, subject to Company performance as measured against financial, operating and strategic objectives, as well as Relative Total Shareholder Return or R-TSR (as defined in Appendix B). The Company’s variable incentives continue to demonstrate a strong linkage between pay and performance.
Annual Cash Incentive
The Company Scorecard (described in more detail on page 53)55) focuses on our most critical business drivers — the Company’s target revenue (“Revenue”), non-GAAP pre-tax income (“Pre-Tax Income”) and, the Company’s research and development goals (“Pipeline”), and the Company’s focus on driving greater access to health for patients around the incentive programworld and on the engagement and inclusion of our employees (“Pipeline”Sustainability”) — and is used to determine the payout of our annual incentive for all eligiblemost employees, including our NEOs under the Executive Incentive Plan. Our Company Scorecard performance during 20212023 resulted in above-target achievement of 148%.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 8 | ||||||||||
ç ç ç ç |
Proxy Summary |
Long-Term Incentive (“LTI”)
The long-term incentive program, consisting of a mix of PSUsPerformance Share Units (“PSUs”) and stock options, provides our NEOs with the opportunity to own Merck common stock, directly linking a substantial portion of their compensation to the returns realized by our shareholders.
The 20192021 PSU program (described in more detail on page 57)58) paid out at 140%161% based on achievement of cumulative two-year OCF, cumulative two-year EPSone-year Earnings Per Share (“EPS”) and three-year R-TSR metrics during the performance period (2021-2023), weighted at 25%, 25%33% and 50%67%, respectively. As previously disclosed, cumulative two-year OCF andone-year EPS metrics werewas used due to the complexities associated with disentangling our Organon & Co. (“Organon”) business from a multi-year financial plan. Organon was successfully spun off in June 2021.
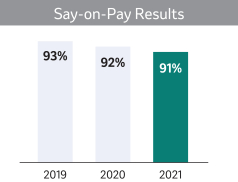
Say-On-Pay Advisory Vote (Page 46)
In 2021, shareholders continued their support for our executive compensation programs with approximately 91% of the votes cast voting in favor of approving the say-on-pay proposal. Consistent with the Company’s strong interest in shareholder engagement and our pay-for-performance approach, the Compensation and Management Development Committee continues to evaluate our executive compensation program to ensure alignment between the respective interests of our executives and shareholders. The C&MD Committee did not make significant changes to our executive compensation program in 2021 as a direct result of the most recent say-on-pay vote.
| Say-On-Pay Advisory Vote (Page 48) | 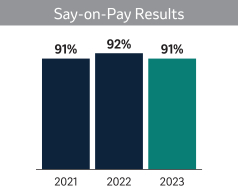 | |
| In 2023, shareholders continued their historically strong support for our executive compensation programs with approximately 91% of the votes cast voting in favor of approving the say-on-pay proposal. Based on this outcome and the Compensation and Management Development (“C&MD”) Committee’s ongoing analysis of the program’s ability to support our strategic, financial, and human capital objectives, we did not make significant changes to our executive compensation program in 2023. Consistent with the Company’s strong interest in shareholder engagement and our pay-for-performance approach, the C&MD Committee continues to evaluate our executive compensation program to ensure alignment between the respective interests of our executives and shareholders. |
We ask that our shareholders approve, on an advisory basis, the compensation of our NEOs as further described in Proposal 2 on page 42.
44. For additional information, please refer to the CD&A beginning on page 4345 of this proxy statement.
Shareholder Engagement and Feedback (Page 24)
Merck communicates regularly with shareholders to better understand their perspectives and has established a shareholder engagement program that is both proactive and cross-functional. In addition, our independent Lead Director, who is also Chair of our Governance Committee, participates in substantive engagements with some of the Company’s largest shareholders. In 2021,2023, discussions with shareholders covered a wide range of topics of interest to shareholders, including the Company’s response to the COVID-19 pandemicsustainability reporting and related matters,goals, the Board’s compositionleadership structure and leadership,composition, management and director succession, Environmental, Social and Governance (“ESG”) reporting, executive compensation programs, human capital management and other governance matters. These discussions provided valuable insights into shareholder views, and we heard from many shareholders that they greatly appreciated the opportunity to engage with our Company.
We will continue to engage with shareholders on a regular basis to better understand and consider their views, including on our sustainability approach, our executive compensation programs ESG and our corporate governance practices.
Board Composition and Refreshment
At least annually, the Governance Committee considers the size, structure and needs of the Board. The Governance Committee reviews possible candidates for the Board and recommends Director nominees to the Board for approval. In addition, as part of the Board’s annual self-evaluation process, Directors provide feedback on Board composition-related matters.
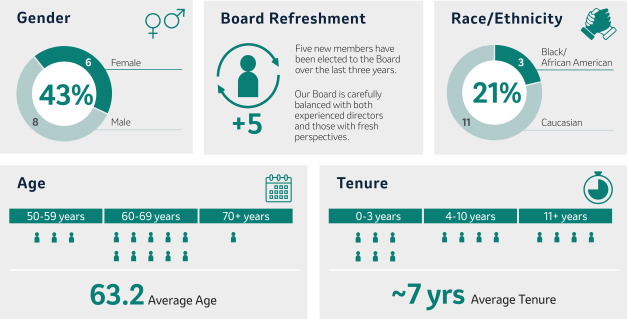
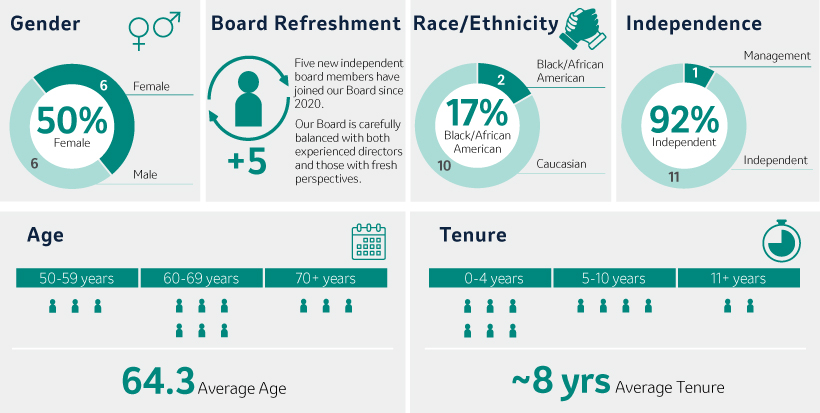
In selecting Director nominees, the Board considers its composition, including its diversity, and the skills, areas of expertise and experience then-represented on the Board. The Board also considers the Company’s current and future global business strategies, opportunities and challenges. Such considerations have resulted in the election of five new Board members over the last three years and the nomination of a new director in this proxy statement.independent Directors since 2020. For more information, see “Criteria“Board Succession Planning, Criteria for Board Membership, and Director Nomination Process” beginning on page 21.
Considering the factors noted above,20. After years of dedicated service, Mr. Peter Wendell will retire from the Board is nominating an additional independent Director to stand for election by shareholders ateffective as of the 20222024 Annual Meeting of Shareholders, Mr. Douglas M. Baker, Jr., Executive Chairman of Ecolab Inc., a provider of water and hygiene services and technologies for the food, hospitality, industrial, and energy markets.Meeting.
Merck & Co., Inc. 2022 2024 Proxy Statement
Proxy Summary | ÷ ÷ ÷ ÷ | 9 |
Nominees for Director (Page 33)36)
The following provides summary information about each Director nominee. Each Director stands for election annually. Detailed information about each individual’s background, skillsets and areas of expertise can be found beginning on page 33.36.
| Current Committee Memberships | ||||||||||||||||||||||||||||||||||
| Audit | C&MD | Governance | Research | |||||||||||||||||||||||||||||||
| Director Nominee | Age | Director Since | Title |  |  |  |  | |||||||||||||||||||||||||||
| Douglas M. Baker, Jr.* | 63 | — | Executive Chairman and Former Chief Executive Officer, Ecolab Inc. | ||||||||||||||||||||||||||||||
| Mary Ellen Coe | 55 | 2019 | President, Google Customer Solutions, Google Inc. |
|
| ||||||||||||||||||||||||||||
| Pamela J. Craig | 65 | 2015 | Former Chief Financial Officer, Accenture plc |
|
| ||||||||||||||||||||||||||||
| Robert M. Davis Management | 55 | 2021 | Chief Executive Officer and President, Merck & Co., Inc. | ||||||||||||||||||||||||||||||
| Kenneth C. Frazier Management | 67 | 2011 | Executive Chairman, Merck & Co., Inc. | ||||||||||||||||||||||||||||||
| Thomas H. Glocer Lead Director | 62 | 2007 | Former Chief Executive Officer, Thomson Reuters Corporation |
|
| ||||||||||||||||||||||||||||
| Risa J. Lavizzo-Mourey, M.D. | 67 | 2020 | Penn Integrates Knowledge Professor of Health Equity and Health Policy, University of Pennsylvania
|
|
| ||||||||||||||||||||||||||||
| Stephen L. Mayo, Ph.D. | 60 | 2021 | Bren Professor of Biology and Chemistry, California Institute of Technology |
|
| ||||||||||||||||||||||||||||
| Paul B. Rothman, M.D.
| 64 | 2015 | Dean of Medical Faculty and Vice President for Medicine, The Johns Hopkins University, and CEO, Johns Hopkins Medicine
|
|
| ||||||||||||||||||||||||||||
| Patricia F. Russo | 69 | 1995 | Chairman, Hewlett Packard Enterprise Company; Former Chief Executive Officer and Director, Alcatel-Lucent
|
|
| ||||||||||||||||||||||||||||
| Christine E. Seidman, M.D. | 69 | 2020 | Thomas W. Smith Professor of Medicine and Genetics, Harvard Medical School, and Director, Cardiovascular Genetics Center, Brigham and Women’s Hospital
|
|
| ||||||||||||||||||||||||||||
| Inge G. Thulin | 68 | 2018 | Former Chairman of the Board, President and Chief Executive Officer, 3M Company
|
|
| ||||||||||||||||||||||||||||
| Kathy J. Warden | 50 | 2020 | Chairman, Chief Executive Officer and President, Northrop Grumman Corporation |
|
| ||||||||||||||||||||||||||||
| Peter C. Wendell | 71 | 2003 | Managing Director, Sierra Ventures |
|
| ||||||||||||||||||||||||||||
Number of Meetings in 2021 | 9 | 5 | 6 | 5 | ||||||||||||||||||||||||||||||
| Current Committee Memberships | |||||||||||||||||||||||||||||||||||||
| Audit | C&MD | Governance | Research | ||||||||||||||||||||||||||||||||||
| Director Nominee | Age | Director Since | Title |  |  |  |  | ||||||||||||||||||||||||||||||
| Douglas M. Baker, Jr. | 65 | 2022 | Founding Partner, E2SG Partners; Former Executive Chairman and Chief Executive Officer, Ecolab Inc. |
|
| |||||||||||||||||||||||||||||||
| Mary Ellen Coe | 57 | 2019 | Chief Business Officer, YouTube Inc. |
|
| |||||||||||||||||||||||||||||||
| Pamela J. Craig | 67 | 2015 | Former Chief Financial Officer, Accenture plc |
|
| |||||||||||||||||||||||||||||||
| Robert M. Davis Management | 57 | 2021 | Chairman, Chief Executive Officer and President, Merck & Co., Inc. | |||||||||||||||||||||||||||||||||
| Thomas H. Glocer Lead Director | 64 | 2007 | Founder/Managing Partner, Angelic Ventures LP; Former Chief Executive Officer, Thomson Reuters Corporation |
|
| |||||||||||||||||||||||||||||||
| Risa J. Lavizzo-Mourey, M.D. | 69 | 2020 | Professor Emerita, Robert Wood Johnson Foundation Population Health and Health Equity, University of Pennsylvania
|
|
| |||||||||||||||||||||||||||||||
| Stephen L. Mayo, Ph.D. | 62 | 2021 | Bren Professor of Biology and Chemistry & Merkin Institute Professor, California Institute of Technology |
|
| |||||||||||||||||||||||||||||||
| Paul B. Rothman, M.D.
| 66 | 2015 | Former Dean of Medical Faculty and Vice President for Medicine, The Johns Hopkins University, and Former CEO, Johns Hopkins Medicine
|
|
| |||||||||||||||||||||||||||||||
| Patricia F. Russo | 71 | 1995 | Former Chief Executive Officer and Director, Alcatel-Lucent
|
|
| |||||||||||||||||||||||||||||||
| Christine E. Seidman, M.D. | 71 | 2020 | Thomas W. Smith Professor of Medicine and Genetics, Harvard Medical School, and Director, Cardiovascular Genetics Center, Brigham and Women’s Hospital
|
|
| |||||||||||||||||||||||||||||||
| Inge G. Thulin | 70 | 2018 | Former Chairman of the Board, President and Chief Executive Officer, 3M Company
|
|
| |||||||||||||||||||||||||||||||
| Kathy J. Warden | 52 | 2020 | Chair, Chief Executive Officer and President, Northrop Grumman Corporation |
|
| |||||||||||||||||||||||||||||||
Number of Meetings in 2023 | 9 | 4 | 4 | 4 | |||||||||||||||||||||||||||||||||
*Mr. Douglas M. Baker, Jr. is a first-time Director nominee for election at the 2022 Annual Meeting of Shareholders. If elected, Mr. Baker would serve as a member of the Compensation and Management Development Committee and the Governance Committee. Effective May 5, 2022, Mr. Baker will retire as Executive Chairman of Ecolab, Inc.
 Committee Chair
Committee Chair
Merck & Co., Inc. 2022 2024 Proxy Statement
| 10 | ||||||||||
ç ç ç ç |
Proxy Summary |
Our 20222024 Director Nominees Snapshot
Our Director nominees possess broad expertise, skills, experience and perspectives that will facilitate the strong oversight and strategic direction required to govern the Company’s business and strengthen and support senior management. As illustrated by the following charts, our slate of Director nominees consists of individuals with expertise in fields that align with the Company’s business and long-term strategy, includes a mixture of tenure that allows for both new perspectives and continuity and reflects the Board’s commitment to diverse perspectives.
Board Skills and Qualifications (of | No. of Nominees* | |||||
CEO Leadership | ||||||
Financial | ||||||
Scientific / Technology | ||||||
Healthcare Industry | 6 | |||||
Global Strategy & Operations | ||||||
Marketing | ||||||
| 7 | |||||
Public Company Governance | ||||||
Public Policy & Regulation | ||||||
Talent Management | ||||||
Capital Markets Experience | 3 | |||||
* See page 22 of this proxy statement under Nominee Skills and Demographic Matrix for how these Board skills and qualifications are represented by each Director nominee individually.
Merck & Co., Inc. 2022 2024 Proxy Statement
Corporate Governance |
| | |||||||
| | |
|
The Board has the legal responsibility for overseeing the affairs of the Company and for the overall performance of the Company. The Board’s primary mission is to represent and protect the interests of our shareholders. To that end, the Board selects and oversees the senior management team, which is charged with conducting Merck’s daily business.
The Board has adopted corporate governance principlesguidelines (the “Policies of the Board”) that, together with our Restated Certificate of Incorporation, By-Laws and Board committee charters, form the governance framework for the Board and its committees. The Policies of the Board cover a wide range of subjects, including the philosophy and functions of the Board, the composition of the Board, the independent Lead Director’s responsibilities, categorical independence standards, Director qualifications, assessment of the Board, committee responsibilities, Director transition and retirement, service on other boards, Director compensation, stock ownership guidelines, chairmanship of meetings, Director orientation and continuing education, incumbent Director resignation and related person transactions. From time to time, the Board revisesupdates the Policies of the Board and Board committee charters in response to changing regulatory requirements, evolving best practices and the perspectives of our shareholders and other constituents.
Most recently, in March 2022, the Board amended: (1) the Policies of the Board and the Charter of the Governance Committee to more expressly reflect (a) the Board’s oversight of ESG matters and (b) the Governance Committee’s assistance in this regard; and (2) the Charter of the Audit Committee to clarify the Audit Committee’s responsibility for reviewing the Company’s cybersecurity risk management program.
Governance Materials
The following items relating to corporate governance at Merck are available on our website at www.merck.com/company-overview/leadership/board-of-directorsboard-of-directors:
| • | By-Laws |
We believe good corporate governance is essential to achieving long-term shareholder value. We are committed to governance policies and practices that serve the interests of our Company and its many stakeholders. For this reason, we devote considerable time and resources to making sure that our policies reflect our values and business goals, we have an effective corporate governance structure, and we operate in an open, honest and transparent way. In addition, we evaluate our practices against prevailing best practices as well as emerging and evolving topics identified in a variety of ways, including through shareholder engagement and corporate governance organizations.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 12 | ||||||||||
ç ç ç ç |
Corporate Governance Board Leadership Structure |
We highlight some significant aspects of our corporate governance practices below.
Independence
• We have a strong independent Lead Director.
• Our independent Directors convene regular executive sessions.
• All four of our standing Board committees (Audit,
•
Accountability
• Every Director stands for re-election every year.
• Directors are elected by majority vote.
• An incumbent director who does not receive a majority vote must tender his/her resignation, and the Governance Committee must promptly make a recommendation as to the tendered resignation. The Board must act on the Governance Committee’s recommendation within 90 days after certification of the vote and publicly disclose its decision and rationale.
Best practices
• Our Board of Directors as a whole, and each
• The Board actively engages in CEO succession planning.
• The Board is diverse in terms of gender, ethnicity,
• Our Board
Transparency
• We have strong control over our political spending and disclose corporate political activity and contributions in the U.S., Canada and Australia.
• We disclose aspects of our public policy engagement, including our key lobbying/advocacy issues. • We disclose philanthropic grants and charitable contributions in the U.S. | Board oversight
• The full Board and each individual Board committee is responsible for overseeing risk.
• The full Board oversees corporate strategy.
Alignment with shareholder interests
• Our officers and directors are prohibited from engaging in hedging, pledging or short sale transactions involving • Executives and Directors must hold prescribed meaningful amounts of • We have a robust shareholder engagement program. • We have a proxy access provision in our By-Laws under which shareholders who own 3% of our outstanding common stock for at least three years may nominate up to 20% of the members of our Board. • Holders of 15% of our shares may call a special meeting. • We do not have a shareholder rights plan (also known as a poison pill). • We do not have any supermajority voting provisions.
Compensation practices
• We have conducted an annual say-on-pay advisory vote since 2011. • All incentive compensation paid to executives is subject to a clawback policy. • Our incentive compensation awards are designed to align pay with performance. • Our
Operating Responsibly
• We have a longstanding commitment to operating responsibly. • All of our employees must adhere to a robust Code of Conduct. |
TheCurrently, the Board is highly empoweredled by Robert M. Davis, who serves as the Chairman of the Board and engaged,CEO of Merck, and by Thomas H. Glocer, an independent Director, who serves as the independent Directors evaluate our Board leadership structure at least annually.Board’s Lead Director. The Board believes that the Company and our shareholders are best served by allowing the Board to exercise its judgment regarding the most appropriate leadership structure for the Company and the Board at a given time. The Board’s discretion should not be unduly constrained in advance because the most appropriate leadership structure at any given time will depend on a variety of factors, including the leadership, skills and experience of each of the CEO, the independent Lead Director and the other members of the Board, as well as the needs of the business and other factors.
Currently, the The independent Directors evaluate our Board is led by Kenneth C. Frazier, who serves as the Executive Chairman of the Board, and by Thomas H. Glocer, an independent Director, who serves as the Board’s Lead Director.leadership structure at least annually.
Merck & Co., Inc. 2022 2024 Proxy Statement
Corporate Governance Lead Director | ÷ ÷ ÷ ÷ | 13 |
The current structure is the result of the Board’s CEO succession planning and regular evaluation of the Company’s leadership structure. In February 2021, the Board unanimously elected Robert M. Davis to succeed Mr. Frazier as CEO and also to become a member of the Board, effective July 1, 2021. The Board also determined that Mr. Frazier will continue to serve as Executive Chairman for a transition period to be determined by the Board. Mr. Glocer was elected independent Lead Director by the independent members of the Board in September 2021 following the resignation of Les Brun due to his decision to become chairman and chief executive officer of Ariel Alternatives, LLC, a subsidiary of Ariel Investments, LLC, a private equity initiative being created to help scale minority-owned businesses to serve as tier 1 suppliers to Fortune 500 companies. Having an independent Lead Director vested with key duties and responsibilities and four independent Board committees chaired by independent Directors promotes strong independent oversight of the Executive Chairman, the CEO and the rest of our management team.
As Executive Chairman, Mr. FrazierDavis presides over meetings of the Board and shareholders and focuses on Board operations and governance matters. He serves as the liaison between the Board and management, working closely with the independent Lead Director and our CEO.Director. Mr. Davis is also in charge of the general supervision, direction and control of the business and affairs of the Company subject to the Board’s overall oversight. The Board meets in executive session without Mr. Frazier and Mr. Davis at each regular Board meeting. During these executive sessions led by theMr. Glocer as independent Lead Director, the Directors discuss topics such as the Board’s leadership structure, succession planning for the CEO and key management positions, and points of follow-up with management on strategic issues.
Merck’s The Board believes that having Mr. Davis serve as Chairman and CEO adds strategic and operational perspective to the Chairman role because he can draw on his detailed institutional knowledge of the Company and industry experience. At the same time, there is strong independent oversight with Mr. Glocer as independent Lead Director and four independent Board committees chaired by independent Directors. Mr. Glocer can communicate with Mr. Davis between meetings and act as a “sounding board” and advisor and is appointedalso vested with key duties and responsibilities as discussed below.
Lead Director
Appointed by the independent members of the Board of Directors, to a three-year term. Thethe position of Lead Director has a clear mandate and significant authority and responsibilities set forth in the Policies of the Board, including:Board. These include:
Board Meetings and Executive Sessions | • The authority to call meetings of the independent members of the Board.
• Presiding at all meetings of the Board at which the | |
Communicating with
| • Serving as the principal liaison on Board-wide issues between the independent members of the Board and the | |
Agendas | • Approving meeting agendas and information sent to the Board, including supporting material for meetings. | |
Meeting Schedules | • Approving meeting schedules to ensure there is sufficient time for discussion of all agenda items. | |
Communicating with Shareholders and Stakeholders | • Being available for consultation and direct communication with major shareholders, as appropriate.
• Serving as a liaison between the Board and shareholders on investor matters. | |
Board Performance Evaluation | • Leading the annual performance evaluation of the Board. | |
Chairman and CEO Performance Evaluations | • Leading the annual performance evaluation of the | |
CEO Succession | • Leading the CEO succession planning process. | |
As further described below,Each of the Board’s four standing committees each of which is composed solely of independent Directors,Directors. As further described below, each of these committees also playplays an active role in the Board’s leadership structure. The independent chairs of each of these committees provide strong leadership to guide the important work of the Board. They work with the Company’s senior executives to ensure the committees are discussing key strategic risks and opportunities of the Company. The Board believes the Company and its shareholders are well-served by the current leadership structure for all the foregoing reasons.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 14 | ||||||||||
ç ç ç ç |
Corporate Governance Board Meetings and Committees |
In
The independent Directors of the Board met in |
All Directors attended at least 75% of the meetings of the Board and of the committees on which they served in |
The Board of Directors has four standing committees, each of which is made up solely of independent Directors: Audit Committee; Compensation and Management Development (C&MD)C&MD Committee; Governance Committee; and Research Committee. In addition, the Board from time to time establishes special purpose committees. All of ourthe standing committees are governed by Board-approved charters, which are available on our website at www.merck.com/company-overview/leadership/board-of-directors/. The committees evaluate their performance and review their charters annually. Additional information about the committees is provided below. As a non-independent directors, Mr. Frazier and director, Mr. Davis areis not membersa member of any Board committee, but may participate in meetings at the request of the committees.
Merck & Co., Inc. 2024 Proxy Statement
Corporate Governance Board Meetings and Committees | ÷ ÷ ÷ ÷ | 15 |
Audit Committee
|
|
| Pamela J. Craig Chair | Overview
The Audit Committee oversees our accounting and financial reporting processes, internal controls and audits and consults with management, the internal auditors, and the independent auditors on, among other items, matters related to the annual audit, the published financial statements and the accounting principles applied. The Audit Committee has established policies and procedures for the pre-approval of all services provided by the independent auditors (as described on page
The Audit Committee’s Report is included on page
The Primary Functions of this Committee are to: • Oversee the Company’s accounting and financial reporting processes, internal controls and audits;
• Appoint, evaluate and retain,
• Oversee the Company’s compliance with legal & regulatory requirements, including monitoring compliance with the Foreign Corrupt Practices Act and the Company’s policies on ethical business practices and reporting on these items to the Board;
• Establish procedures for the receipt, retention and treatment, on a confidential basis, of
• Oversee the Enterprise Risk Management process;
• Regularly meet with the Chief Information Officer regarding the Company’s information technology and have primary responsibility for overseeing the Company’s cybersecurity risk management program; and
• Review any significant issues concerning litigation and contingencies with management, counsel, and the independent public accountants. | |||||||
Other Members
Stephen L. Mayo, Ph.D. Paul B. Rothman, M.D. Christine E. Seidman, M.D. Kathy J. Warden
| |||||||||
Number of Meetings in 9
| |||||||||
Financial Experts on Audit Committee
The Board has determined that each of Mr. Baker, Ms. Craig and Ms. Warden is an “audit committee financial expert” as defined by the SEC and has accounting or related financial management expertise as required by NYSE Corporate Governance Listing Standards.
| |||||||||
Merck & Co., Inc. 2022 Proxy Statement
|
|
Compensation and Management Development Committee
|
|
| Patricia F. Russo Chair | Overview
The C&MD Committee annually reviews and approves corporate goals and objectives relevant to the
The C&MD Committee Report is included on page
The Primary Functions of this Committee are to:
• Establish and maintain a competitive portfolio of fair and equitable compensation and benefits policies, practices and programs designed to attract, engage and retain a workforce that helps the Company achieve immediate and long-term success;
• Discharge the Board’s responsibilities for compensating our officers;
• Oversee/monitor – The competence and qualifications of our executive officers, – Officer succession, – The soundness of the organizational structure, – The Company’s programs, policies and practices related to its management of human capital resources including talent management, culture, diversity, equity and inclusion, and provide input on the same, and – Other related matters necessary to ensure the effective management of the business; and
• Review the Compensation Discussion and Analysis for inclusion in our proxy statement. | |||||||
Other Members Mary Ellen Coe Thomas H. Glocer Risa J. Lavizzo-Mourey, M.D. Inge G. Thulin Peter C.
| |||||||||
Number of Meetings in
| |||||||||
Compensation and Management Development Committee Interlocks and Insider Participation
There were no C&MD Committee interlocks or insider (employee) participation during
| |||||||||
(1) Mr. Wendell is retiring from the Board effective as of the 2024 Annual Meeting.
Merck & Co., Inc. 2024 Proxy Statement
| 16 | ç ç ç ç | Corporate Governance Board Meetings and Committees |
Governance Committee
|
|
| Thomas H. Glocer Chair | Lead Director
| Overview
The Governance Committee oversees the Company’s corporate governance, including the practices, policies and procedures of the Board and its committees. Further, the Governance Committee annually reviews the size, structure and needs of the Board and Board committees, reviews possible candidates for the Board and recommends Director nominees to the Board for approval. The details of the review process and assessment of candidates are described under
The Primary Functions of this Committee are to:
• Coordinate an annual evaluation of Board performance, and review Board compensation, related person transactions and
• Oversee the Board’s Incumbent Director Resignation Policy;
• Review the
• Review social, political and economic trends that affect our business; review the positions and strategies we pursue to influence public policy; and
• Assist the Board in its oversight of the Company’s | |||||||
Other Members Douglas M. Baker, Jr. Pamela J. Craig Patricia F. Russo Inge G. Thulin Kathy J. Warden
| |||||||||
Number of Meetings in
| |||||||||
(1) Ms. Warden was appointed to the Governance Committee as of March 22, 2022. She previously served on the Research Committee.
Merck & Co., Inc. 2022 Proxy Statement
| ||||||||
Research Committee
|
|
| Paul B. Rothman, M.D. Chair | Overview
The Research Committee oversees the overall strategy, direction and effectiveness of the
The Primary Functions of this Committee are to:
• Identify areas and activities that are critical to the success of our product and vaccine
• Keep the Board apprised of this evaluation process and findings and make appropriate
• Assist the Board in its oversight responsibilities to ensure compliance with the highest | |||||||
Other Members Mary Ellen Coe Risa J. Lavizzo-Mourey, M.D. Stephen L. Mayo, Ph.D. Christine E. Seidman, M.D. Peter C. Wendell(1)
| |||||||||
Number of Meetings in
| |||||||||
(1) Dr. Lavizzo-Mourey was appointed toMr. Wendell is retiring from the Research CommitteeBoard effective as of March 22, 2022. She previously served on the Governance Committee.2024 Annual Meeting.
Merck & Co., Inc. 2022 2024 Proxy Statement
Corporate Governance Board’s Role in Strategic Planning | ÷ ÷ ÷ ÷ | 17 |
Board’s Role in Strategic Planning
The Board — acting both as a whole and through its four standing committees — is fully engaged and involved in the Company’s strategic planning process. All of our Directors have an obligation to keep informed about the Company’s business and strategies, so they can provide guidance to management in formulating and developing plans and knowledgeably exercise their decision-making authority on matters of importance to the Company.
The Board’s oversight and guidance are inextricably linked to the development and review of the Company’s strategic plan. By exercising sound and independent business judgment on the strategic issues that are important to the Company’s business, the Board facilitates Merck’s long-term success.
Our Strategic Planning Cycle


Merck & Co., Inc. 2022 2024 Proxy Statement
| 18 | ||||||||||
ç ç ç ç |
Corporate Governance Risk Oversight |
Overseeing risk is an important component of the Board’s engagement on strategic planning. The Board’s approach to overseeing risk management leverages the Board’s leadership structure and ensures the Board oversees risk through both a Company-wide approach and specific areas of competency. A summary of this risk oversight approach follows:
Board of Directors
Oversees risk through Company-wide Enterprise Risk Management (“ERM”) process and functioning of Board Committees. |
Audit Committee
Responsibility for reviewing the ERM process to ensure it is robust and functioning effectively.
Primary responsibility for overseeing the Company’s cybersecurity risk management
Oversees risk relating to finance, business integrity and
| |||
Compensation
Evaluates relationships between risk and
When setting incentive plan targets each year, the C&MD Committee is aware of the risk associated with drug pricing, among other things, and ensures our plans do not incentivize risky behavior in order to meet targets.
Oversees the Company’s programs, policies and practices related to its management of human capital resources.
| ||||
Management
Identification, assessment and management of risk through Company-wide ERM process. | ||||
Governance Committee
Oversees the Company’s corporate governance, including the practices, policies and procedures of the Board and its committees, considers the size, structure and needs of the Board, reviews possible candidates for the Board, and recommends Director nominees to the Board for approval.
Plays a role in compliance oversight, including in the areas of manufacturing quality, privacy, and worker safety.
Assists the Board in its | ||||
Research Committee
Oversees overall strategy, direction and effectiveness of the Company’s research and development operations.
|
The ERM process allows for full Board oversight of the most significant risks facing the Company and was established to ensure a complete Company-wide approach to evaluating risk over six distinct but overlapping risk areas:
Responsibility and Reputation | Risks that may impact the well-being of the Company, its employees, customers, patients, communities or reputation | ||
Strategy | Macro risks that may impact our ability to achieve long-term business objectives | ||
Operations | Risks in operations and cybersecurity that may impact our ability to achieve business objectives | ||
Compliance | Risks related to compliance with laws, regulations and Company values, ethics and policies | ||
Reporting | Risks to maintaining accurate financial statements and timely, complete financial disclosures | ||
Safety | Risks to employee, patient or community health and safety | ||
Merck & Co., Inc. 2022 2024 Proxy Statement
Corporate Governance Risk Oversight | ÷ ÷ ÷ ÷ | 19 |
Our ERM process seeks to identify emerging risks and address them appropriately to limit negative consequences to the Company orand the data it maintains. Its goal is to provide an ongoing review, implemented across the Company and aligned to Company values and ethics, to identify and assess risk and to monitor risk and agreed-upon mitigating action. Furthermore, if a risk transforms into an incident, the ERM process ensures that effective response and business continuity plans are in place. If the ERM process identifies a material risk, it will be elevated through the CEO and the Executive Team to the full Board for consideration. Through the ERM process, each Board committee oversees specific areas of risk relevant to the committee through direct interactions with the CEO, members of the Company’s Executive Team and the heads of business divisions, compliance and corporate functions. A committee may address risks directly with management or, where appropriate, may elevate a risk for consideration by the full Board or another Board committee. The Board committees also oversee risk based on their specific areas of competency. Additional detail with respect to certain key areas of oversight are provided below.
As our Company becomesdiscussed in more dependentdetail in the 2023 10-K, the Company’s cybersecurity measures are primarily focused on ensuring the security and protection of its information technology systems and data,data. The Audit Committee and the need for a robustBoard receive periodic reports that include updates on the Company’s cybersecurity privacy,risks and technology riskthreats, the status of projects intended to strengthen its information security systems, assessments of the information security program (including remediation, mitigation, and management of identified vulnerabilities), and the emerging threat landscape. The Company’s information security program is increasingly critical. We have developedregularly evaluated by internal and implemented a comprehensive program designedexternal consultants and auditors with the results of those reviews reported to protect the confidentiality of sensitive information, ensure the integrity of critical data and automated processes, and safeguard the availability of our information technology capabilities.
Cybersecurity has been an area ofsenior management attention for over two decades and we have aligned our cybersecurity program to the National Institute of Standards and Technology (NIST) Cybersecurity Framework and the Payment Card Industry Data Security Standard (PCI-DSS). We have implemented appropriate policies, processes, and technology to reduce the likelihood or impact of a breach and have cyber insurance. We have an employee awareness program to regularly educate our workforce on the cybersecurity risks they face and how they can operate safely. We regularly assess our cybersecurity capabilities using third party security firms including an annual assessment of our adherence to the PCI-DSS standard.Audit Committee.
We have also developed and continually evolve our Global Privacy Program to promote organizational accountability for privacy, data governance, and data protection across our business and with our collaborative partners and suppliers. The program helps us uphold our commitment to data security and privacy, including maintaining 100% compliance to regulatory requirements for active incident monitoring, risk/harm analysis, and on-time notification of data breaches. Our commitment applies not only to our Company’s information, but also to the information entrusted to us by others. We were the first company in the world to obtain regulatory certification in the European Union for Binding Corporate Rules based in part on our existing Asia Pacific Economic Cooperation Cross Border Privacy Rules certification.
We are aware that we must continuously evolve our controls to address new threats, adhere to changing laws and standards, and reduce the risk associated with the introduction of new, innovative technology.
While everyone at the Company plays a part in information security, cybersecurity, and data privacy, oversight responsibility is shared by the Board, its committees, and management.
Responsible Party | Oversight Area for Cybersecurity and Privacy Matters | |
Board | Participates in | |
Audit Committee |
| |
Governance | Responsible for oversight in the area of privacy and receives periodic updates regarding the Company’s Global Privacy Program. | |
Management | Responsible for implementing and managing the Company’s framework for assessing, prioritizing and mitigating cybersecurity risk. A Chief Information Security Officer manages the Company’s Information Security Program with a group responsible for leading enterprise-wide cybersecurity risk management, strategy, policy, standards, architecture and processes. Manages the Company’s Global Privacy Program. Responds to incidents and issues in a timely manner. Provides periodic updates to the Board and or its committees, as applicable. |
Merck & Co., Inc. 2022 2024 Proxy Statement
| 20 | ||||||||||
ç ç ç ç |
Corporate Governance
Board Succession Planning, Criteria for Board Membership, and Director Nomination Process |
Environmental, Social and Governance (“ESG”) MattersSustainability
The work to address our environmental footprintBoard of Directors and social impact begins with the Board, which as a wholeits Committees are responsible and through its committees, has responsibilityaccountable for overseeing the Company’s ESG matters. In general:sustainability matters, and management is responsible for reviewing, refining, and implementing the long-term sustainability strategy and for updating the Board and its Committees.
Responsible Party | Key Oversight | |
Board |
| |
Governance |
| |
|
| |
Audit Committee |
| |
Research Committee |
| |
Management |
By ensuring ongoing business engagement ownership and accountability with regard to sustainability, management is working to create long-term value, to differentiate our Company as a leader, and to respond to stakeholder requests for |
A Strategic ApproachFor more information about our approach to ESGsustainability, please visit our 2022/2023 Impact Report athttps://www.merck.com/company-overview/sustainability/.
Our ESG strategy applies our global resourcesBoard Succession Planning, Criteria for Board Membership, and investmentsDirector Nomination Process
In Board succession planning, the Governance Committee and the Board consider, among other things, the needs of the Board and the Company in light of the overall composition of the Board, with a view toward achieving a balance of the skills, experience and attributes that are essential to the priority areas that matter mostBoard’s oversight role. In particular, the Board is deliberate in ensuring the Board has the right mix of diverse perspectives, skills and expertise to societyaddress the Company’s current and anticipated needs as opportunities and challenges facing the Company evolve. The Board also strives to our business:
Access to Health
Employees
Environmental Sustainability
Ethics & Values
Addressing these focus areas enables us to better reach thoseensure an appropriate mix of tenure on the Board and being balanced with both experienced and newer directors. Such considerations have resulted in need with our medicines and vaccines; to help to build robust, durable health systems worldwide; to developthe election of five new independent Board members since 2020. The Board also has a diverse, inclusive and healthy global workforce; to demonstrate our environmental stewardshippolicy set forth in our operations and in our supply chain; to enhance our relationships with key stakeholders; and to continue to contribute toward the UN Sustainable Development Goals (“SDGs”). This commitment to a high level of ESG performance helps to drive sustainable value for our business and for society.
Advancing Health Equity is Key to our Access Strategy
As a research-intensive biopharmaceutical company, our role is to discover and develop innovative medicines and vaccines that address unmet medical needs and somePolicies of the world’s greatest health threats. AsBoard that Directors may not be nominated for re-election to the Board after they reach the age of 75. The Board believes that, in addition to its ongoing review of the overall composition of the Board, this policy promotes regular refreshment of the Board and is considered as part of this pursuit, we have a responsibility to an increasingly diverse set of patients, including addressing the systemic issues that have long impacted underrepresented groups.
Our global approach to Access to Health is one of our four ESG focus areas. Our strategy is driven by our Access to Health Guiding Principles, which span the areas of discovery and invention, availability, affordability, and strengthening systems and addressing inequity. In addressing access, we are focused on improving people’s health, contributing to social and economic development and helping to achieve the United Nations’ Sustainable Development Goal for Good Health and Well-Being (SDG 3) through our health equity efforts.overall succession planning.
Merck & Co., Inc. 2022 2024 Proxy Statement
Corporate Governance Board Succession Planning, Criteria for Board Membership, and Director Nomination Process | ÷ ÷ ÷ ÷ | 21 |
Pursuing health equity, the idea that everyone has a fair and just opportunity to be as healthy as possible, is key to our access strategy. Addressing preventable differences in the burden of disease and health outcomes with people who are underserved can make a meaningful, measurable and lasting impact in patients’ lives.
In December 2021, we further underscored our legacy of catalyzing solutions that improve health equity by announcing an additional $150 million commitment through Merck for Mothers. This program promotes safe, high-quality, and respectful maternal care in underserved communities, and we aim to reach 25 million women through these initiatives and services by 2025. Merck for Mothers will continue to focus efforts on countries where the need is great, including India, Nigeria, Kenya and the U.S., bringing on new collaborators and strategic investments that further scale the impact of the current 100+ programs across 50+ global sites.
We also pursue strategies to provide direct access to our products to those who need them. A recent example are the non-exclusive voluntary licensing agreements we have entered into with the Medicines Patent Pool and established generic manufacturers. We aim to accelerate and facilitate affordable global access to molnupiravir—our investigational oral antiviral COVID-19 medicine—in more than 100 low- and middle-income countries following approvals or emergency authorization by local regulatory agencies.
When market-based solutions are inadequate or unavailable, we also pursue programs to provide direct access to our medicines and vaccines, including product donations and patient assistance programs. This includes our longstanding support of the Mectizan® Donation Program (“MDP”). The MDP provides our Company’s product Mectizan—as much as needed, for as long as needed—with the goal to help eliminate river blindness and lymphatic filariasis and provide equitable solutions in remote communities where health services are limited.
Through these initiatives, we continue to challenge ourselves to innovate, make ambitious commitments, and form collaborative partnerships to advance health equity.
$1 Billion Bond Issued to Support ESG Strategy
In December 2021, we announced a $1 billion sustainability bond to support eligible projects and partnerships, including:
Access to essential health care services (affordability and addressing barriers to health, infectious disease R&D)
Socioeconomic advancement and empowerment (employee/supplier diversity and inclusion, health literacy)
Renewable energy generation
Energy efficiency
Green buildings
Sustainable water and wastewater management
Pollution prevention and control
For more information on this bond, please see our Company’s Investor Relations page at www.merck.com/investor-relations/.
Criteria for Board Membership and Director Nomination Process
The Governance Committee is responsible for screening and nominating director candidates to be considered for election by the Board. As part of this process, the Governance Committee considers the composition of the Board at the time, including the depth of experience, balance of professional skills, expertise and diversity of perspectives represented by its members at the time. The Governance Committee evaluates prospective nominees identified on its own initiative as well as candidates recommended by other Board members, management, shareholders or search consultants. In 2021,
Diversity
As a Company, Merck knows that diversity and inclusion are fundamental to the Company’s success and core to future innovation. As a Board, diversity is an important factor considered when identifying prospective nominees for our Board, and the Policies of the Board include a formal diversity policy. The policy reflects the Board’s longstanding commitment to ensuring that Directors represent diverse perspectives and areas of expertise important to fostering the Company’s business success. The policy provides that the Board does not discriminate against potential Directors on the basis of gender, race, age, sexual orientation or ethnic and national background and that having a board composed of diverse individuals is an important contributor to the Board’s overall effectiveness.
Shareholder Recommendations of Director Candidates
The Governance Committee retained a search firmwill consider recommendations for Director candidates made by shareholders and will evaluate those individuals using the same criteria applied to identify possible candidates who meetother candidates. Shareholder recommendations must be sent to the Board’s qualifications, to interview and screen such candidates (including conducting reference checks) and to assist in scheduling candidate interviews with Board members.
Office of the Secretary, Merck & Co., Inc. 2022 Proxy Statement
Candidates are evaluated initially based on materials submitted by them or on their behalf. If a proposed or recommended candidate continues to be of interest to the Governance Committee, we obtain additional information through inquiries to various sources and, if warranted, interviews.
|
Individual Experience, Qualifications, Attributes and Skills
In its regular discussions regarding Board composition — and especially in conjunction with the annual Board and committee evaluations — the Governance Committee works with the Board to determine the appropriate mix of professional experience, expertise, educational background and other qualifications that are particularly desirable in light of our current and future business strategies. The Governance Committee uses this input in its planning and Director search process.
To be considered for membership on the Board, a candidate must meet the following minimum criteria:
be of proven integrity with a record of substantial achievement in an area of relevance to the Company;
have demonstrated ability and sound judgment that usually will be based on broad experience;
be able and willing to devote the required amount of time to the Company’s affairs, including attendance at Board meetings, Board committee meetings and annual shareholder meetings;
possess a judicious and critical temperament that will enable objective appraisal of management’s plans and programs; and
be committed to building sound, long-term Company growth.
Individual Experience, Qualifications, Attributes and Skills
In its regular discussions regarding Board composition — and especially in conjunction with the annual Board and committee evaluations — the Governance Committee works with the Board to determine the appropriate mix of professional experience, expertise, educational background and other qualifications that are particularly desirable in light of our current and future business strategies. The Governance Committee uses this input in its planning and Director search process. In addition to the five broadthese criteria, listed above, the following chartmatrix highlights the background, experiencemix of key skills and skillsqualifications the Board considers for future candidates. These attributes are amply represented by our current Director nominees.
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
Diversity
As a Company, Merck knows that diversity and inclusion are fundamental to the Company’s success and core to future innovation. As a Board, diversity Additional biographical information on each nominee is an important factor considered when identifying prospective nominees for our Board, and the Policies of the Board include a formal diversity policy. The policy reflects the Board’s longstanding commitment toset out starting on page 36.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 22 | ç ç ç ç |
Corporate Governance
Nominee Skills and Demographic Matrix |
ensuring that Directors represent diverse perspectivesThe following biographical disclosures include information regarding nominees for election at the 2024 Annual Meeting only. After years of dedicated service, Peter Wendell will retire as of, and areas of expertise important to fosteringnot stand for reelection at, the Company’s business success. The policy provides that2024 Annual Meeting, and is not represented in the Board does not discriminate against potential Directors on the basis of gender, race, age, sexual orientation or ethnicfollowing biographical disclosures.
Nominee Skills and national background and that having a board composed of diverse individuals is an important contributor to the Board’s overall effectiveness.Demographic Matrix
Shareholder Recommendations of Director Candidates
Skills and Experience of our 2024 Director Nominees |  |  |  |  |  |  |  |  |  |  |  |  | ||||||||||||
CEO Leadership Experience serving as a chief executive officer at a publicly traded or private organization | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||
Financial Experience or expertise in financial accounting and reporting processes or the financial management of a major organization | ● | ● | ● | ● | ● | ● | ||||||||||||||||||
Scientific / Technology Scientific or technological degree or work experience in a scientific or technological field | ● | ● | ● | ● | ● | ● | ||||||||||||||||||
Healthcare Industry Experience with complex issues within the healthcare industry | ● | ● | ● | ● | ● | ● | ||||||||||||||||||
Global Strategy & Operations Leadership experience overseeing and/or driving strategic direction and growth of an organization globally | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
Marketing / Sales Experience in marketing, advertising, social media and consumer insight functions, including product development and brand building | ● | ● | ● | ● | ||||||||||||||||||||
Digital Experience or expertise in information technology (including cybersecurity and data privacy) or the use of digital media to facilitate business objectives | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||
Public Company Governance Experience as a board member of another publicly-traded company | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||
Public Policy & Regulation Experience with public policy and regulation in the healthcare industry or other highly- regulated industries | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||
Talent Management Experience in executive recruiting, succession planning and talent management, including retaining key talent and driving employee engagement | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
Capital Markets Experience Experience in corporate lending or borrowing, capital market transactions, significant mergers or acquisitions, private equity or investment banking | ● | ● | ● | |||||||||||||||||||||
Certain Demographic Information | ||||||||||||||||||||||||
Tenure (years) | 2 | 5 | 9 | 3 | 17 | 4 | 3 | 9 | 29 | 4 | 6 | 4 | ||||||||||||
Age | 65 | 57 | 67 | 57 | 64 | 69 | 62 | 66 | 71 | 71 | 70 | 52 | ||||||||||||
Gender | M | F | F | M | M | F | M | M | F | F | M | F | ||||||||||||
Race/Ethnicity | ||||||||||||||||||||||||
Black/African American | ● | ● | ||||||||||||||||||||||
White | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
The Governance Committee will consider recommendations for Director candidates made by shareholders and will evaluate those individuals using the same criteria applied to other candidates. Shareholder recommendations must be sent to the Office of the Secretary,
Merck & Co., Inc., 2000 Galloping Hill Road, K1-4157, Kenilworth, New Jersey 07033 U.S.A., and must include detailed background information regarding the recommended candidate that demonstrates how that candidate meets the Board membership criteria. 2024 Proxy Statement
Corporate Governance Management Succession Planning | ÷ ÷ ÷ ÷ | 23 |
Candidates are evaluated initially based on materials submitted by them or on their behalf. If a proposed or recommended candidate continues to be of interest to the Governance Committee, we obtain additional information through inquiries to various sources and, if warranted, interviews.
Management Succession Planning
Succession planning and talent development are important at all levels within the Company. The Board regularly reviews short- and long-term succession plans for the CEO and other executive officers. In assessing possible CEO candidates, the independent Directors identify the skills, experience and attributes they believe are required for an effective CEO in light of the Company’s global business strategies, opportunities and challenges. More broadly, the Board engages with the Company’s leadership team on matters of talent and culture, including around the development of the Company’s talent pipeline and advancing diversity and inclusion efforts across the enterprise. The Board’s succession planning activities are strategic, long-term and supported by the Board’s committees and external consultants, as needed, and Directors have substantial opportunities to engage with possible succession candidates. Most recently, this succession planning process resulted in a new leadership structure for the Company’s Human Health business to enable the Company to develop our internal talent and expand our high performing senior leadership team following the departure of Frank Clyburn, former Executive Vice President and President, Human Health. Effective February 28, 2022, Arpa Garay leads Human Health Global Marketing, Jannie Oosthuizen leads Human Health U.S., and a third role will lead Human Health International. The Board provided guidance regarding the new leadership structure. The Board also approved the appointments of Ms. Garay and Mr. Oosthuizen and will be responsible for approving the appointment of the third role.
The Board also considers its own composition and succession plans. In Director succession planning, the Governance Committee and the Board consider, among other things, the needs of the Board and the Company in light of the overall composition of the Board, with a view toward achieving a balance of the skills, experience and attributes that are essential to the Board’s oversight role. In particular, the Board is deliberate in ensuring the Board has the right mix of diverse perspectives, skills and expertise to address the Company’s current and anticipated needs as opportunities and challenges facing the Company evolve. Such considerations have resulted in the election of five new Board members over the last three years and the nomination in this proxy statement of a new Director. In addition, in November of 2021, the Board amended the Policies of the Board to provide that Directors may not be nominated for re-election to the Board after they reach the age of 75. The Board believes that, in addition to its ongoing review of the overall composition of the Board, this policy promotes regular refreshment of the Board and is considered as part of overall succession planning.
Merck & Co., Inc. 2022 Proxy Statement
|
The Board conducts an evaluation of its performance and effectiveness, as well as that of its four standing committees, on an annual basis. The purpose of the evaluation is to track progress in certain areas targeted for improvement, identify ways to enhance the overall effectiveness of the Board and its committees, track progress in certain areas targeted for improvement, and provide opportunities to discuss other important topics. Among other topics, each Director provides feedback on the following topics that the full Board then considers:
the Board’s effectiveness in evaluating and monitoring the Company’s strategy and risks;
whether the Board has the right mix of skills, experiences, and attributes in light of the current issues facing the Company and its strategy for the future;
whether the Board’s leadership structure remains appropriate and effective;
evaluating such Director’s contributions as well as the contributions of other Directors; and
whether there is adequate time to raise questions, and make comments, both during and between Board meetings.
In addition, each Committee member evaluates their respective Committees, including with respect to leadership, composition, succession plans and priority agenda topics.functioning. The independent Lead Director leads the evaluation process. The Governance Committee may also from time to time engage an independent third party to manage the process. In 2021,2023, the evaluation was conducted in 3 phases.phases, and led to refinements to meeting agendas, streamlining of materials, and identification of priority agenda topics for the Board to address in 2024.

Merck & Co., Inc. 2024 Proxy Statement
| 24 | ||||||||||||||||||
ç ç ç ç |
Shareholder Engagement and Feedback | |||||||||||||||||
 |  |  | ||||||||||||||||
|
Shareholder Engagement and Feedback
Merck regularly communicates with shareholders to better understand their perspectives and has established a shareholder engagement program that is proactive and cross-functional. Throughout the year, members of Investor Relations, the Office of the Secretary, Human Resources and the ESG Strategy and Engagement Team, as well as other subject-matter experts within the Company, engage with our shareholders to remain well-informed regarding their perspectives on current issues and to address any questions or concerns. These teams serve as liaisons between shareholders, members of senior management and the Board. We also regularly seek to take advantage of other engagement opportunities and events.
In addition, we conduct an extensivea formal shareholder outreach program twice a year focused on governance, executive compensation and ESG matters.sustainability. We believe it is most productive to discuss these matters well in advance of the Company’s Annual Meeting of Shareholders to enable management and the Board to gather information about investorshareholder perspectives and make educated and deliberate decisions that are balanced and appropriate for Merck’s diverse shareholder base and in the Company’s best interests. Given our large shareholder base, we concentrate our formal outreach efforts on those of our largest 3050 shareholders, whichwho have stewardship teams involved in proxy voting. Our largest 50 shareholders represented approximately 40%50% of our ownership as of December 31, 2021,2023, based on filings made by our shareholders with the SEC on or before March 1, 2022.5, 2024.
During 2021,
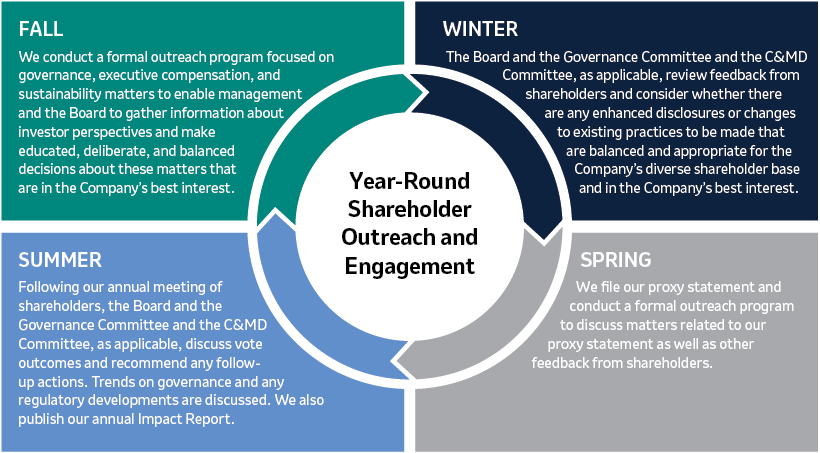
Merck & Co., Inc. 2024 Proxy Statement
Corporate Governance Shareholder Communications with the Board | ÷ ÷ ÷ ÷ | 25 |
In 2023, specifically, we followed this process and held discussions with a number of our shareholders in the spring before the Annual Meeting of Shareholders that year and once again in late fall. Our independent Lead Director, who is also Chair of the Governance Committee, participated in substantive engagements with some of the Company’s shareholders. We also regularly seek to take advantage of other engagement opportunities and events.
Merck & Co., Inc. 2022 Proxy Statement
|
|
Topics Discussed with Shareholders during | ||||
• Company strategy • Board leadership, composition and refreshment • Management succession • Board and management diversity • Human capital management • • Shareholder proposals
| • Global access to Merck products • • Risk oversight • Cybersecurity • Executive compensation programs • Policy and pricing environment • |
• Climate initiatives • Merck Animal Health • Director tenure • Merck culture • Reputation •
| ||
Some key themes emerged as partWe heard from a number of shareholders that they appreciated the Company’s disclosures of certain demographic information about Directors but also expressed interest in understanding this information on an individualized basis. Following discussion with the Governance Committee and the Board, we added a matrix on page 22 providing demographic information regarding each Director nominee on an individualized basis.
In addition, we heard that shareholders appreciated the Company’s existing disclosures regarding access and pricing transparency for our various engagements as set forth below.medicines and vaccines, but some were interested in additional disclosure focused on the Company’s approach to patents and access. We included such disclosure on pages 26 to 30.
 What We Heard What We Heard |  What We Did What We Did | |
|
| |
|
|
Proxy Access
After engaging with a number of our largest shareholders, our Board proactively amended our By-Laws in 2015 to give shareholders a right to proxy access for Director nominations. Our By-Laws allow a shareholder (or a group of no more than twenty shareholders) who has maintained continuous qualifying ownership of at least 3% of the Company’s outstanding common stock for at least three years to include Director nominees constituting up to 20% of the Board in the Company’s proxy materials for an annual meeting of shareholders. Our By-Laws, which prescribe additional requirements for proxy access, are available on our website at www.merck.com/company-overview/leadership/board-of-directors/.
Merck & Co., Inc. 2022 Proxy Statement
|
Shareholder Communications with the Board
The Board of Directors welcomes input from shareholders and other interested parties and has established a process to receive these communications. Shareholders and interested parties may communicate directly with the Board, the independent Lead Director, the non-management or independent Directors as a group or other members of the Board by emailing office.secretary@merck.com, or by writing to the following address:
Board of Directors,
Merck & Co., Inc.
2000 Galloping Hill Road, K1-4157
Kenilworth, NJ 07033 126 East Lincoln Avenue, Rahway, N.J. 07065 U.S.A.
In order to manage efficiently the volume of correspondence received, communications will be reviewed by the Office of the Secretary for the purpose of determining whether the contents are appropriate for submission to the entire Board, the Chairman, the independent Lead Director or the Chair of a particular committee. The Office of the Secretary will not transmit:
communications that advocate that the Company engage in illegal activity;
communications that, under community standards, contain offensive or abusive content;
communications that have no relevance to the role of the Board or to the business of the Company;
resumes or other job-related inquiries; and
| • | resumes or other job-related inquiries; and |
mass mailings, solicitations and advertisements.
Merck & Co., Inc. 2024 Proxy Statement
| 26 | ç ç ç ç | Corporate Governance Access and Pricing Transparency for Our Medicines and Vaccines |
Comments or questions regarding the nomination of Directors and other corporate governance matters will be referred to the Chair of the Governance Committee. Comments or questions regarding executive compensation will be referred to the Chair of the C&MD Committee.
In addition, the Audit Committee has established procedures for the receipt, retention and treatment, on a confidential basis, of complaints regarding accounting, internal accounting controls or auditing matters, and the confidential, anonymous submissions by employees of concerns regarding questionable accounting or auditing matters. These procedures are described in the Merck Code of Conduct — Our Values and Standards.
The Merck Code of Conduct is available on our website atwww.merck.com/company-overview/culture-and-values/code-of-conduct/values-and-standards/.
Access and Pricing Transparency for Our Medicines and Vaccines
Our Company
At Merck, our mission is to use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have worked to help solve the world’s toughest medical challenges. We have developed essential childhood vaccines, introduced the first protease inhibitor, which helped transform AIDS from a death sentence to a chronic disease, and developed the first statin to demonstrate a significant benefit on health outcomes from reducing high cholesterol. More recently, we have been working on the treatment and prevention of cancer. For example, we have been researching and developing KEYTRUDA (pembrolizumab), the Company’s anti-PD-1 (programmed death receptor-1) therapy. Since its first approval in 2014, our Company has demonstrated the efficacy of KEYTRUDA in 39 indications across 17 tumor types and 2 tumor agnostic indications and reached nearly 2 million patients. The impact of KEYTRUDA and other recent treatment advances is difficult to overstate, with a recent American Cancer Society report finding that cancer mortality in the United States has fallen 33% from 1991 to 2021, representing an estimated 4 million Americans whose deaths have been averted.1
Our product GARDASIL is the first FDA-approved vaccine to guard against the human papillomavirus (HPV). HPV is the leading cause of the vast majority of cases of cervical cancer, the fourth most common cancer of women globally. A study of real world evidence published in the New England Journal of Medicine evaluating data from Swedish girls and women between 10 and 30 years of age found a substantially reduced risk of invasive cervical cancer among those who had been fully vaccinated with GARDASIL.2 An American Cancer Society report found similar transformative public health outcomes in the United States, with a 65% decrease in cervical cancer rates in women in their early 20s, following the widespread adoption of HPV vaccines in the United States.3
Our Commitment
We continue to focus on innovative health solutions that advance disease prevention and treatment for both humans and animals and are committed to operating responsibly. This commitment has remained a pillar of strength for us, forming the foundation of our strategic framework and business approach. We are dedicated to putting patients first, upholding ethical standards, demonstrating integrity, showing respect for people, and driving innovation and scientific excellence across all aspects of our operations.
In 2023, we reached more than 500 million people with our innovations across commercial channels, clinical trials, access strategies like our partnership with Gavi and UNICEF, voluntary licensing of antiviral treatments including our investigational antiviral COVID-19 medicine, as well as medicine and vaccine donations.
| 1 | Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA: A Cancer J Clin. https://doi.org/10.3322/caac.21820. Published Jan. 17, 2024. Accessed Feb. 3, 2024. |
| 2 | Lei J, Ploner A, Elfstrom E, et al. HPV Vaccination and the Risk of Invasive Cervical Cancer. N Engl J Med 2020; 383:1340-1348. DOI: 10.1056/NENMoa1917338. |
| 3 | Siegel RL, Miller KD, Wagle NS, et al. Cancer statistics, 2023. CA: A Cancer J Clin. https://doi.org/10.3322/caac.21763. Published Jan. 12, 2023. Accessed Feb. 3, 2024. |
Merck & Co., Inc. 2024 Proxy Statement
Corporate Governance Access and Pricing Transparency for Our Medicines and Vaccines | ÷ ÷ ÷ ÷ | 27 |
We generally report progress on our company-wide Access to Health goals on an annual basis. Our current goals are to:
Reach at least 75 percent of countries in the world annually with our products
Enable access for 350 million more people to our innovative medicines and vaccines through access strategies, solutions, and partnerships by 2025
| • | Advance health equity by reaching 50 million people in low- and middle-income countries (LMICs) and populations underserved by healthcare in high-income countries, by 2025 |
Information regarding our Company’s progress against these goals is available in our most recent annual Impact Report: https://www.merck.com/wp-content/uploads/sites/124/2023/08/Merck-Impact-Report-22-23.pdf.
Pricing Transparency
We have a long history of making our medicines and vaccines accessible and affordable through responsible pricing practices and industry-leading patient-access programs. We are continuing to work to help patients overcome access and affordability challenges.
To provide information about our pricing practices, we have posted on our website an annual Pricing Transparency Report for the United States. The report provides the average annual list price, net price increases and average discounts across the Company’s U.S. portfolio dating back to 2010.
Access & Regulatory
To advance our goal of enabling more people to access our innovative portfolio, we are focusing on building healthcare capacity, strengthening channels for care delivery and fostering sustainable financing. In building healthcare capacity, we help to create greater efficiency for patients in the healthcare system through collaborations with hospitals and healthcare networks. For example, we work with partners using a data-based approach to understand how a patient’s cancer journey can be optimized from earlier diagnosis through to treatment. We also collaborate with different financial institutions and payers, supporting them to expand funding options that help patients and their families cope with potentially high out-of-pocket costs related to critical illness. Through collaborations that reach underserved populations, we are working with healthcare providers and others in the digital health and financial sectors to develop solutions that enhance disease awareness and healthcare access.
We seek to ensure global access to our medicines and vaccines by obtaining and maintaining up-to-date product registrations around the globe. In order to make our products available to the people who need them throughout the world, we registered 156 products and devices in 2022, the majority of which were registered in LMICs in the Asia-Pacific, Central and Eastern Europe, Middle East and Africa, and Americas regions.
We have also taken steps to address the unique needs of LMICs where the infrastructure and personnel necessary to deliver immunization services can be severely limited. Specifically, we have focused on product improvements such as the introduction of vaccine vial monitors for use to assess controlled temperature-chain conditions. In May 2022, we obtained World Health Organization approval for our HPV vaccines’ compatibility for use outside the cold chain for up to four days. This helps enhance accessibility for hard-to-reach populations.
Patient Assistance Program
When market-based solutions are inadequate or unavailable, we pursue programs to provide direct access to our medicines and vaccines, including product donations and patient assistance programs. Individuals who cannot afford our medicines and have no other means of coverage, public or private, may be eligible to be provided with our medicines, at no charge. In the past five years, our patient assistance program in the United States has helped nearly 800,000 patients to obtain our medicines or vaccines free of charge, with an estimated value of $7.8 billion.4
| 4 | Periods prior to June 2021 include products included in spin-off to Organon & Co. |
Merck & Co., Inc. 2024 Proxy Statement
| 28 | ç ç ç ç |
Corporate Governance Access and Pricing Transparency for Our Medicines and Vaccines |
Patent Exclusivity and Access
Innovation and access to health are central to our Company’s purpose as a research-intensive biopharmaceutical company. Our Access to Health Guiding Principles, available at https://www.merck.com/wp-content/uploads/sites/124/2021/08/Merck-Access-to-Health-Principles_Update-2021.pdf, guide our global approach to access to health.
We believe intellectual property is an enabler and not a barrier to access to innovative, quality-assured medicines and vaccines. Strong intellectual property systems are the cornerstone of innovation, catalyzing the investment needed to embark on the long, costly, and high-risk process of developing new and improved medicines and vaccines. Intellectual property systems not only reward innovation, but also allow scientists to build on each other’s discoveries and provide a framework for collaboration.
In the pharmaceutical industry, a product’s market exclusivity is generally determined by two forms of intellectual property rights: patents owned or controlled by the innovator company and any forms of regulatory exclusivity to which the innovative medicine is entitled. Patents provide our Company with the right to exclude others from practicing an invention for a limited time. Without a meaningful period of market exclusivity, our Company’s ability to invest in research and development (R&D) to discover and develop promising new medicines may be limited.
The extensive R&D process typically leads to numerous types of innovations associated with a medicine. Patents related to a commercial product may cover, among other things, the product’s active ingredient(s), different uses of the product (such as methods of treating a disease), pharmaceutical formulations directed to specific compositions of active and inactive ingredients within the product, delivery mechanisms, and processes for (or intermediates useful in) the manufacture of the product.
We believe it is important to protect our Company’s ability to innovate by continuing to file for core intellectual property rights that protect important innovations. Because of how the patent system is structured, a patent application typically needs to be filed before the Company will know whether the disclosed invention will be relevant to a product candidate in development, or even to a marketed product, or can effectively evaluate the patient-access situation for the disclosed invention. Often, after a related patent application is filed, years of clinical development are still needed to determine a product candidate’s safety and efficacy and whether or not to pursue regulatory approval and what the access situation may be if regulatory approval were to be pursued and received. As such, our Company evaluates all patent applications and resulting patents annually to assess whether they should be maintained.
Our Company conducts a fact-specific and complicated scientific and legal analysis of new innovations discovered during the R&D process to determine whether or not to pursue patent protection. We evaluate our innovations for potential patenting using similar processes deployed by patent offices when they examine the merits of applications for patentability. Each potential patent application is evaluated separately, including the timing of when and where to file on a particular innovation.
Each patent application is subject to rigorous review by individual patent offices in the countries and/or regions in which it is filed. A patent is granted only when determined to meet the standards of patentability in that country or region. Protection afforded by patents may vary from country to country and depends upon the type of patent and its scope of coverage. In the 2023 10-K, we disclosed the various estimated minimum patent exclusivity dates for each of our key products in the U.S., EU (as specifically represented by France, Germany, Italy and Spain), Japan and China, which is typically the expiration of the patent that claims the active ingredient of the product.
A patent directed to the active ingredient(s) of a potential drug product is typically the first application associated with the potential product to be filed, before any clinical testing occurs. Continued investment in R&D occurs through the drug development process, and often also occurs after the drug product is first commercialized to explore additional potential benefits for patients. For example, additional research may demonstrate that a medicine can also be used to treat different states of the same disease, such as earlier stages of cancer, or to treat completely different conditions, including different forms of cancer or different diseases altogether. In other cases, additional research may lead to a demonstration of increased safety or effectiveness of the medicine, new dosage forms, or new forms of administration of a medicine that can improve patient adherence or convenience, leading to better patient outcomes.
By way of example, since 2014, when KEYTRUDA was first approved to treat advanced or unresectable melanoma, our Company has worked tirelessly to investigate its use in treating additional types of cancers. The FDA requires innovators to
Merck & Co., Inc. 2024 Proxy Statement
Corporate Governance Access and Pricing Transparency for Our Medicines and Vaccines | ÷ ÷ ÷ ÷ | 29 |
prove their products work by conducting clinical trials for patients having each distinct type of cancer. Each of these clinical trials can take several years to conduct, and there is no guarantee of success. While more than 45 pivotal trials studying KEYTRUDA produced results that enabled the approvals of KEYTRUDA described above, there are more than 25 pivotal trials studying KEYTRUDA that did not produce results supportive of approval.
Between 2011, when our focused KEYTRUDA research program began, and 2023, Merck has invested approximately $30 billion in our own internal clinical development efforts, $14 billion in research collaborations and acquisitions to further the study of KEYTRUDA with other compounds, and $2 billion in capital expenditures to scale up our processes and facilities to manufacture the medicine in large quantities. We expect to invest another $18 billion in KEYTRUDA clinical studies into the 2030s.5
Patents play a key role in safeguarding the investments made in researching and developing additional uses, and our Company prioritizes obtaining intellectual property for those innovations that provide the greatest medical benefit to patients. Importantly, however, patents directed to, for example, additional indications or combination treatments (so-called “secondary or tertiary” patents), which might be added to a product’s label in the future, do not extend the patent life of earlier patents covering the initial innovation associated with a product. This is because each patent has its own respective exclusivity period, which is limited to the specific scope of the patent’s claims. To bring its version of KEYTRUDA to the market, a biosimilar manufacturer does not need to “copy” the subsequent innovation that our Company has made around KEYTRUDA and protected with patents. The FDA enables biosimilar manufacturers to seek approval for fewer than all conditions of use for which the innovator product has been approved. To this end, we continue to expect manufacturers will offer biosimilar options for that initial discovery of pembrolizumab, the active ingredient in KEYTRUDA, by late 2028 when the patent covering the pembrolizumab antibody structure expires.
Access Planning
We build on our legacy of putting patients first by inventing medicines and vaccines to address areas of unmet medical need. This is evident in our strategic priorities that include investing in, augmenting and accelerating our pipeline to deliver life changing products and demonstrating value to our stakeholders and extending access to solutions that address unmet medical needs.
Embedded within our R&D process, we systematically evaluate our candidates to identify the potential to address significant public health burden and unmet medical needs in underserved healthcare settings. This evaluation process informs our product access strategies, with the goal of making our medicines and vaccines available to as many people as possible through sustainable solutions.
To facilitate access, we undertake a systematic evaluation at the onset of Phase 2 clinical studies to determine a candidate’s potential to address unmet medical needs in LMICs. For candidates with significant potential in LMICs as well as settings with underserved populations,6 access planning may start in the pre-clinical phase. Additionally, understanding where health system infrastructure and funding mechanisms are in place is an important component of enabling safe and effective usage, which ultimately facilitates meaningful patient access.
Our R&D Governance Committee is accountable for the evaluation process, and all recommendations are reviewed by our Strategic Policy and Sustainability Council, an internal cross-divisional forum of senior leaders. When a medicine or vaccine candidate with the potential to address significant public health burden in underserved healthcare settings is identified, the access planning process includes engaging all parts of our enterprise, as well as external stakeholders, to identify the optimal solution.
Once approved, we commit to making the product available in all countries where clinical trials have been conducted. Products continue to be evaluated for their potential to address disease burden throughout their life cycle to account for changes in the external environment.
Sometimes the evaluation of a candidate during the R&D process reveals barriers to access in LMICs or underserved settings. In these situations, the evaluation process can inform our approach to strengthening health systems and improving health equity. We recognize that addressing the complex and multi-faceted challenges to accessing healthcare in LMICs requires the collaboration of multiple stakeholders. We actively seek partnerships to achieve solutions that enable access.
| 5 | Figures in this paragraph are rounded. |
| 6 | Underserved populations are defined as those that face health disparities due to disadvantages related to insurance status, social determinants of health, race, ethnicity, gender identity/sexual orientation, age and/ or language preference. |
Merck & Co., Inc. 2024 Proxy Statement
| 30 | ç ç ç ç | Corporate Governance Political Contributions and Lobbying Expenditure Oversight and Disclosure |
For our manufacturing efforts, this means we are balancing internal and external licensing opportunities, including assessing new voluntary licensing and technology transfer strategies and expanding our network for supply chain design. This business strategy enables enterprise-level strategic segmentation, highlighting the specific access gaps in markets and guiding us to commercially sustainable solutions, including new archetypes for manufacturing and supply execution. Our manufacturing division engages early in the lifecycle of a program through a new product development and commercialization process to understand and facilitate future manufacturing, supply chain and network needs. While not all elements will be known, an early concept of the supply and manufacturing strategy will be critical to preserve future options. This means we start supply chain design early in Phase 1 of clinical development.
Extending access to innovations that address unmet medical need is considered when deciding how to exercise our legitimate intellectual property rights. Future access to our Company’s new medicines and/or new images or uses is considered at multiple stages in a product’s life cycle, including as it approaches the regulatory approval process, after approval of the medicine, and during litigation or any licensing around the product. We only obtain intellectual property by lawful and ethical means, and only enforce those intellectual property rights we believe to be valid. While we continue to believe it is important to protect innovation with intellectual property rights, as a product life-cycle develops, we are deeply committed to making decisions about enforcement or licensing of those rights in a manner consistent with our mission and commitment, including impact on patient access. This may include choosing not to file or enforce our patents in certain parts of the world, partnering with multilateral organizations, non-governmental organizations, and governments to enable greater access in certain markets, particularly in LMICs, and providing financial or free product support through a comprehensive global network of patient assistance programs to qualified patients where allowed by local law. We also support and participate in fair dealing with respect to arms-length, commercial patent settlements entered into with generic manufacturers.
Political Contributions and Lobbying Expenditure Oversight and Disclosure
Merck is committed to participating constructively and responsibly in the political process and to providing clarifying analysis and information regarding the issues that affect our business and patient care. The Company advocates for public policies that foster research into innovative medicines and improve access to medicines, vaccines and health care.healthcare. Our participation in the political process is guided by the following principles: improving patient access to healthcare, including access to medicines and vaccines, improving access to animal health products, and encouraging innovation. The Company’s public policy positions are determined by senior management with oversight by the Governance Committee. Our political contributions are made in accordance with all applicable laws and Company policies and procedures and are overseen by senior management. The Governance Committee monitors all such contributions, and the full Board receives a bi-annual report.
In addition, the Company publicly discloses and regularly updates information regarding its public policy positions and advocacy expenditures on our website at www.merck.com/company-overview/responsibility/transparency-disclosures/. This information includes the Company’s contributions, categorized by state, candidate and amount, for our corporate political and political action committee contributions in the U.S., Canada and Australia. These disclosures include information for the past 5 years. In addition, this information includes a list of U.S. industry and trade groups in which we are members where our dues are greater than $25,000 and the portion of our dues that these groups use for advocacy and/or political activities.
Governance and Transparency around Drug Pricing
In order to provide information about the Company’s pricing practices, the Company annually posts on its website its Pricing Transparency Report for the United States. The report provides the Company’s average annual list price, net price increases and average discounts across the Company’s U.S. portfolio dating back to 2010. In 2021, the Company’s gross U.S. sales were reduced by 43.5% as a result of rebates, discounts and returns. Our process around pricing our products includes regular presentations to the Board on drug pricing strategies. In addition, on balance, over the last few years, our revenue growth has been primarily attributable to increased volume arising from increased demand for our products rather than price increases.
The Policies of the Board require that a substantial majority of our Directors be independent. In making independence determinations, the Board observes all relevant criteria established by the U.S. Securities and Exchange Commission (the “SEC”) and the New York Stock Exchange (the “NYSE”), as well as categorical independence standards set forth in the Policies of the Board. The Board considers all relevant facts and circumstances in making an independence determination.
To be considered independent, an outside director must meet the bright line independence tests established by the NYSE, and the Board must affirmatively determine that the director has no direct or indirect material relationship with the Company.
Merck & Co., Inc. 2024 Proxy Statement
Corporate Governance Related Person Transactions | ÷ ÷ ÷ ÷ | 31 |
The Board also rigorously considers all relevant heightened independence requirements for members of the Audit Committee and the C&MD Committee. The Governance Committee reviews the Board’s approach to determining director independence periodically and recommends changes, as appropriate, for consideration and approval by the full Board.
Independence Determinations
In accordance with the NYSE Corporate Governance Listing Standards and the categorical standards reflected in the Policies of the Board, the Board reviewed relationships between the Company and each Director. As a result of that review, the Board has determined that, with the exception of Robert M. Davis, our Chairman, CEO and President, and Kenneth C. Frazier, our Executive Chairman, each Director has only immaterial relationships with the Company, and accordingly, each is independent under these standards. The Board also has determined that each member of the Audit Committee, the C&MD Committee
Merck & Co., Inc. 2022 Proxy Statement
|
and the Governance Committee is independent within the meaning of the NYSE Corporate Governance Listing Standards and the rules of the SEC.
In making these determinations, the Board considered relationships that exist between the Company and other organizations where Directors serve, as well as the fact that in the ordinary course of business, transactions may occur between such organizations and the Company or one of our subsidiaries. The Board also evaluated whether there were any other facts or circumstances that might impair a Director’s independence.
Drs. Lavizzo-Mourey Rothman and Seidman are employed at medical or academic institutions with which the Company engages in purchase and/or sale transactions in the ordinary course of business. Ms. Coe is employed by Google Inc., and, in 2020, the Company engaged in a purchase transaction with Google Inc. in the ordinary course of business. In addition, Mr. Thulin was employed by 3M Company until June 1, 2019, and the Company engages in routine business transactions with 3M Company. The Board reviewed transactions with each of these entities and determined that the applicable individual Director had no role with respect to the Company’s decision to make any of the purchases or sales, and the aggregate amounts in each case were less than 2% of the consolidated gross revenues of the other organization and the Company.
Related Person Transaction Policy
The Board has adopted a written Related Person Transaction Policy (the “Policy”) that is incorporated into the Policies of the Board and administered by the Governance Committee. The Policy governs the review and approval of any transactions involving amounts exceeding $120,000 to which the Company or a subsidiary is a party and in which a “related person” has a direct or indirect material interest. A “related person” is any Director, Director nominee, executive officer or holder of more than 5% of any outstanding class of the Company’s voting securities, as well as immediate family members or certain affiliated entities of any of the foregoing persons.
Pursuant to the Policy, management determines whether a transaction requires review by the Governance Committee, in which case the transaction, along with all material information, will be disclosed to the Governance Committee for review, approval, ratification or termination. In the event a related person transaction is approved by the Governance Committee, such transaction will be subject to ongoing monitoring to ensure that the transaction remains fair and reasonable to the Company. For additional information, the full Policy is available on the Company’s website atwww.merck.com/company-overview/leadership/board-of-directors/in the Policies of the Board.
Certain Related Person Transactions
Each Director and executive officer of Merck annually, and each Director nominee before such nominee’s nomination, completes and submits to the Company a Director & Officer (“D&O”) Questionnaire. The D&O Questionnaire requests, among other things, information regarding whether any Director, Director nominee, executive officer or their immediate family members had an interest in any transaction or proposed transaction with Merck or its subsidiaries or has a relationship with a company that has entered or proposes to enter into such a transaction.
After review of the D&O Questionnaires by the Office of the Secretary, the responses are collected, summarized and distributed to responsible areas within the Company to identify any potential transactions. All relevant relationships and any transactions, along with payables and receivables, are compiled for each person and affiliation. Management submits a report of the affiliations, relationships, transactions and appropriate supplemental information to the Governance Committee for its review. Based on this information for 2021,2023, the Governance Committee has determined that no transactions require disclosure under Item 404(a) of SEC Regulation S-K.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 32 | ç ç ç ç |
Corporate Governance Compensation Consultants |
Role of Compensation Consultants
The C&MD Committee retains the services of a compensation consultant to serve as an objective third-party advisor on the reasonableness of compensation levels and on the appropriateness of the compensation program structure in supporting our business strategy and human resource objectives. Since 2008, the C&MD Committee has retained FW Cook as its compensation consultant. In addition, the Governance Committee periodically retainshas retained, and may in the future retain FW Cook to assist with a review of the Directors’ compensation program.
Independence of the Compensation Consultant
The C&MD Committee annually reviews the services provided by FW Cook and has concluded that FW Cook is independent in providing executive compensation consulting services. The C&MD Committee conducted a specific review ofevaluated its relationship with FW Cook in 2021,2023 and determined that FW Cook’s work for the C&MD Committee did not raise any conflicts of interest, consistent with the guidance provided under the Dodd-Frank Act and by the SEC and the NYSE, determined that FW Cook’s work for the C&MD Committee did not raise any conflicts of interest.NYSE. In making this determination, the C&MD Committee reviewed information provided by FW Cook on the following factors:
In particular, the C&MD Committee noted that (i) FW Cook provided no other services to Merck; and (ii) FW Cook’s work is performed directly on behalf of the Board working in cooperation with management, to assist both the C&MD Committee and the Governance Committee, as applicable, with executing their respective responsibilities.
Services Performed During 20212023
During 2021,2023, FW Cook supported the C&MD Committee by:in the following areas:
Since 2010, management has retained Pay Governance LLC to provide consulting services on an as-needed basis. Although Pay Governance did not provide any services during 2021,2023, they will perform their biennial risk assessment of our compensation programs in November 2022.2024.
Merck & Co., Inc. 2022 2024 Proxy Statement
| ÷ ÷ ÷ ÷ | 33 |
Stock Ownership of Directors and Officers
The table below reflects the number of shares of Merck common stock beneficially owned by (a) each of our Directors; (b) each of our executive officers named in the Summary Compensation table; and (c) all Directors and executive officers as a group. These numbers are rounded to the nearest whole share. As of February 28, 2022, 2,527,811,51229, 2024, 2,533,298,070 shares of Merck common stock were issued and outstanding. Unless otherwise noted, the information is stated as of February 28, 2022,29, 2024, and the beneficial owners exercise sole voting and/or investment power over their shares. In addition, unless otherwise indicated, the address for each person named below is c/o Merck & Co., Inc., 2000 Galloping Hill Road, Kenilworth,126 East Lincoln Avenue, Rahway, New Jersey 07033.07065.
| Company Common Stock | ||||||||||||||||
Name of Beneficial Owner | Shares Beneficially Owned(1) | Right to Acquire Beneficial Exercisable/Distributable Within 60 Days(2) | Percent of Class | Phantom Stock Units(3) | ||||||||||||
Kenneth C. Frazier | 710,665 | 3,668,920 | * | — | ||||||||||||
Robert M. Davis | 247,593 | 430,029 | * | — | ||||||||||||
Mary Ellen Coe | 10 | — | * | 12,949 | ||||||||||||
Pamela J. Craig | 1,715 | — | * | 20,624 | ||||||||||||
Thomas H. Glocer | 5,100 | — | * | 80,578 | ||||||||||||
Risa J. Lavizzo-Mourey | 1,000 | — | * | 5,486 | ||||||||||||
Stephen L. Mayo | 100 | — | * | 2,911 | ||||||||||||
Paul B. Rothman | 100 | — | * | 20,625 | ||||||||||||
Patricia F. Russo | 13,148 | — | * | 44,787 | ||||||||||||
Christine E. Seidman | 100 | — | * | 7,164 | ||||||||||||
Inge G. Thulin | 100 | — | * | 14,043 | ||||||||||||
Kathy Warden | 500 | — | * | 5,610 | ||||||||||||
Peter C. Wendell | 1,000 | — | * | 111,095 | ||||||||||||
Frank Clyburn(4) | 98,875 | 263,812 | * | 2,241 | ||||||||||||
Richard R. DeLuca, Jr. | 135,063 | 370,888 | * | — | ||||||||||||
Dean Li | 15,462 | 49,723 | * | — | ||||||||||||
Caroline Litchfield | 26,556 | 147,787 | * | — | ||||||||||||
All Directors and Executive Officers as a Group (28 individuals) | 1,591,687 | 5,769,706 | * | 342,916 | ||||||||||||
| Company Common Stock | ||||||||||||||
Name of Beneficial Owner | Shares Beneficially Owned(1) | Right to Acquire Beneficial Ownership Under Options/Stock Units Exercisable/Distributable Within 60 Days(2) | Percent of Class | Phantom Stock Units(3) | ||||||||||
Robert M. Davis | 348,327 | 275,441 | * | — | ||||||||||
Douglas M. Baker, Jr. | 1,000 | — | * | 4,502 | ||||||||||
Mary Ellen Coe | 10 | — | * | 20,822 | ||||||||||
Pamela J. Craig | 1,715 | — | * | 26,323 | ||||||||||
Thomas H. Glocer | 5,100 | — | * | 93,087 | ||||||||||
Risa J. Lavizzo-Mourey | 1,000 | — | * | 10,306 | ||||||||||
Stephen L. Mayo | 100 | — | * | 7,582 | ||||||||||
Paul B. Rothman | 100 | — | * | 26,323 | ||||||||||
Patricia F. Russo | 13,148 | — | * | 51,887 | ||||||||||
Christine E. Seidman | 100 | — | * | 12,859 | ||||||||||
Inge G. Thulin | 100 | — | * | 19,360 | ||||||||||
Kathy Warden | 500 | — | * | 10,437 | ||||||||||
Peter C. Wendell(4) | 1,000 | — | * | 124,525 | ||||||||||
Sanat Chattopadhyay | 157,192 | 232,394 | * | 14,803 | ||||||||||
Richard DeLuca | 137,013 | 212,224 | * | — | ||||||||||
Dean Li | 46,242 | 145,789 | * | — | ||||||||||
Caroline Litchfield | 50,908 | 192,933 | * | — | ||||||||||
All Directors and Executive Officers as a | 926,956 | 1,411,677 | * | 422,816 | ||||||||||
| * | Less than 1% of the Company’s outstanding shares of common stock. |
| (1) | Includes equivalent shares of common stock held by the Trustee of the Merck U.S. Savings Plan, for the accounts of individuals as follows: Mr. |
| (2) | This column reflects the number of shares that could be acquired within 60 days of February |
| (3) | Represents phantom shares denominated in Merck common stock under the Plan for Deferred Payment of Directors’ Compensation or the Merck Deferral Program. |
| (4) | Mr. |
Merck & Co., Inc. 2024 Proxy Statement
| 34 | ç ç ç ç | Stock Ownership Information Delinquent Section 16(a) Reports |
Delinquent Section 16(a) Reports
Section 16(a) of the Exchange Act, as amended, requires our executive officers, directors, and beneficial owners of more than 10% of our common stock to file stock ownership reports and reports of changes in ownership with the SEC. Based on a review of those reports and written representations from the reporting persons, to our knowledge, all such reports for 2021
Merck & Co., Inc. 2022 Proxy Statement
|
|
2023 were filed on a timely basis, except: (1)except one Form 3/A by Chirfi Guindo filed on April 28, 2023 solely to restate the number of shares beneficially and directly owned by Mr. Guindo, and one Form 4 by Richard R. DeLuca, Jr. reporting a special one-time Restricted Stock Unit retention grant due on May 6, 2021, butJoseph Romanelli filed on November 4, 2021, (2) one Form 4June 30, 2023 solely to report purchases and sales that were not directed by Frank Clyburn also reportingMr. Romanelli, but were rather executed by a special one-time Restricted Stock Unit retention grant due on May 6, 2021, but filed on November 4, 2021, and (3) one Form 4 by David M. Williams reportingbrokerage firm for the vestingbenefit of Restricted Stock Units due on February 12, 2021, but filed on February 11, 2022.Mr. Romanelli’s managed account without his knowledge.
Stock Ownership of Certain Beneficial Owners
The table below reflects the number of shares beneficially owned by persons or entities known to us to own more than 5% of the outstanding shares of Merck common stock as of December 31, 2021.2023. As of December 31, 2021, 2,527,604,6292023, 2,531,633,273 shares of Merck common stock were issued and outstanding.
Name and Address of Beneficial Owner | Amount and Nature of Beneficial Ownership | Percent of Class | |||||||||||
The Vanguard Group 100 Vanguard Blvd., Malvern, PA 19355 | |||||||||||||
BlackRock, Inc. 55 East 52nd Street, New York, NY 10055 | |||||||||||||
| (1) | As reported on Amendment No. |
| (2) | As reported on Amendment No. |
Merck & Co., Inc. 2022 2024 Proxy Statement
Election of Directors | | | | | |
35 | ||||||||||
| ||||||||||||
The Board has recommended 1412 nominees for election as Directors at the 20222024 Annual Meeting of Shareholders:Meeting: Mr. Douglas M. Baker, Jr., Ms. Mary Ellen Coe, Ms. Pamela J. Craig, Mr. Robert M. Davis, Mr. Kenneth C. Frazier, Mr. Thomas H. Glocer, Dr. Risa J. Lavizzo-Mourey, Dr. Stephen L. Mayo, Dr. Paul B. Rothman, Ms. Patricia F. Russo, Dr. Christine E. Seidman, Mr. Inge G. Thulin, and Ms. Kathy J. Warden and Mr. Peter C. Wendell.Warden. All nominees, other than Mr. Davis, our Chairman and Chief Executive Officer, and Mr. Frazier, our Executive Chairman, satisfy the NYSE independence requirements.
In connection with the Board’s CEO succession planning, in February 2021, Mr. Peter Wendell will be retiring from the Board elected Mr. Davis, effective July 1, 2021, to succeed Mr. Frazier as CEO and become a member of the 2024 Annual Meeting. Mr. Wendell has a distinguished history of insightful contributions since joining the Board in 2003, and stand for election by shareholders at the 2022 Annual Meeting of Shareholders. Mr. Baker is being nominated to stand for election by shareholders at the meeting. Board has benefited from his extensive management, financial and venture capital expertise.
All otherDirector nominees currently serve on the Board and were elected by the shareholders at the 20212023 Annual Meeting. Mr. Baker was first identified as a possibleMeeting of Shareholders.
In addition, all Director candidate by a third-party search firm. Mr. Baker was recommended to the Board by the Governance Committee and met with various members of the Board leading up to his nomination.
All Director nominees named in this proxy statement meet the Board’s criteria for membership and were recommended by the Governance Committee, and approved by the Board, for election by shareholders at the 20222024 Annual Meeting. All of them hold, or have held, senior leadership positions in large, complex organizations, including multi-national corporations, medical or academic institutions, or charitable organizations. In these positions, our Director nominees have demonstrated their leadership, intellect and analytical skills and gained deep experience in core disciplines significant to their oversight responsibilities at Merck.the Company. Their varied roles and experiences reflect a diversity of perspectives, skills and expertise to address the Company’s current and anticipated needs as the Company’s opportunities and challenges evolve. If elected, each nominee will serve until the 20232025 Annual Meeting of Shareholders or until a successor has been duly elected and qualified, subject to their earlier resignation, death or removal.
Any Director nominee who does not receive a majority of the votes cast with respect to his or her election will not be re-elected as a Director of the Company. However, under the New Jersey Business Corporation Act, incumbent Directors who are not re-elected in an uncontested election because of a failure to receive a majority of the votes cast in favor of their re-election will be “held over” and continue as Directors of the Company until they resign, or their successors are elected at the next election of directors. Our Incumbent Director Resignation Policy, included in the Policies of the Board, provides that an incumbent Director who is not re-elected must promptly submit a resignation. The Governance Committee will evaluate whether to accept such resignation and make a recommendation to the full Board. The Board must act on the recommendation no later than 90 days following certification of the shareholder vote and publicly disclose its decision and rationale.
If any Director nominee becomes unavailable for election (which we do not expect), votes will be cast for such substitute Director nominee or nominees as may be designated by the Board, unless the Board reduces its size.
There are no family relationships among Merck’sthe Company’s executive officers and Directors.
We provide below biographical information for each Director nominee, including key experience, qualifications and skills such Director nominee contributes to the Board in light of our current needs and business priorities.
FOR
The Board of Directors recommends that the shareholders vote FOR the election of each of the Director Nominees.
|
Merck & Co., Inc. 2022 2024 Proxy Statement
| 36 | ç ç ç ç |
Proposal 1 Election of Directors |
|
Douglas M. Baker, Jr. Independent
|
| ||||||||||
Age:
| ||||||||||||
Director
| ||||||||||||
Committees:
| ||||||||||||
 |  | |||||||||||
|
Governance | |||||||||||
Experience
|
Mr. Baker has wide-ranging expertise in corporate governance and general and organizational management, including a deep understanding of global marketing, sales and operations of public companies. |
Career Highlights
|
E2SG Partners • Founding Partner (2022-Present) Ecolab Inc. • Executive Chairman • Chairman and Chief Executive Officer (2006-2020) • Chief Executive Officer (2004-2006)
|
Other Public Directorships
|
Current •
Former • Ecolab Inc. (2006-2022) • U.S. Bancorp (2008-2018)
|
|
|
|
Mary Ellen Coe Independent
|
| ||||||||||
Age:
| ||||||||||||
Director Since: 2019
| ||||||||||||
Committees:
| ||||||||||||
 |  | |||||||||||
|
Research
| |||||||||||
Experience
|
Ms. Coe has a deep understanding of the digital, media and technology landscape, as well as global strategy and operations, due to her experience as a senior leader at YouTube Inc. and Google Inc. At YouTube, Ms. Coe currently serves as the Chief Business Officer, leading all global business operations, content plus distribution partnerships across multiple areas and YouTube’s monetization ecosystem, including streaming businesses like YouTube TV, YouTube Music and YouTube Premium, in addition to supporting the company’s advertising business. At Google, Ms. Coe previously served as the President of Google Customer Solutions, overseeing the global ads business for mid-market and small businesses, serving millions of customers and thousands of partners worldwide. She also has extensive marketing and sales expertise from her leadership position at McKinsey and other global marketing consulting firms. |
Career Highlights
|
YouTube Inc. • Chief Business Officer (2022-Present) Google Inc. • President, Google Customer Solutions • Vice President, Go-to-Market Vice President, Go-to-Market Operations and Strategy (2012-2017)
|
Other Public Directorships
|
Current • None Former • Whole Foods Market, Inc. (2016-2017)
|
Merck & Co., Inc. 2022 2024 Proxy Statement
Proposal 1 Election of Directors | ÷ ÷ ÷ ÷ | 37 |
|
Pamela J. Craig Independent
|
| ||||||||||||
Age:
| ||||||||||||||
Director Since: 2015
| ||||||||||||||
Committees:
| ||||||||||||||
 |  | |||||||||||||
Audit (Chair)
|
Governance
| |||||||||||||
Experience
|
Ms. Craig has extensive finance, management, operational, technology and international business expertise, including her history of accomplishment and executive ability as Chief Financial Officer of Accenture plc. In addition, her directorships at other public companies, including her service as a member of the Audit Committee and |
Career Highlights
|
Accenture plc, global management consulting, technology services and outsourcing company • Chief Financial Officer (2006-2013) • Senior Vice President, Finance • Group Director, Business Operations and Services (2003-2004) • Managing Partner, Global Business Operations (2001-2003)
|
Other Public Directorships
|
Current • Progressive Insurance (since 2018) •
Former • 3M Company (2019-2023) • Akamai Technologies, Inc. (2011-2019) • Wal-Mart Stores, Inc. (2013-2017)
|
|
Robert M. Davis Management
|
| ||||||||
Age:
| ||||||||||
Director Since: 2021
| ||||||||||
| ||||||||||
| ||||||||||
Experience
|
Mr. Davis, Merck’s Chairman, Chief Executive Officer and President, has extensive management, financial, and operational expertise. |
Career Highlights
|
Merck & Co., Inc. • Chairman (2022-present) • Chief Executive Officer and President (2021-present) • Chief Financial Officer and Executive Vice President, Global Services (2016-2021) • Chief Financial Officer and Executive Vice President (2014-2016) Baxter International, Inc. • Corporate Vice President and President, Medical Products (2010-2014) • Corporate Vice President and Chief Financial Officer (2006-2010) • Corporate Vice President and Treasurer (2004-2006)
|
Other Public Directorships
|
Current • Duke Energy (since 2018) Former • C.R. Bard (2015-2017)
|
Merck & Co., Inc. 2022 2024 Proxy Statement
| 38 | ç ç ç ç |
Proposal 1 Election of Directors |
|
|
| ||||||||
Age:
| ||||||||||
Director Since: | ||||||||||
|
|
|
|
|
|
| ||||||||||
| ||||||||||
| ||||||||||
Committees:
| ||||||||||
 |  | |||||||||
Compensation and Management Development
|
Governance (Chair) | |||||||||
| ||||||||||
Experience
|
Mr. Glocer has extensive management, operational, technology and international business expertise, including his history of accomplishment and executive ability as CEO and a Director of Thomson Reuters Corporation. In addition, his directorships at other public companies, including his service as Lead Director and as a member of the |
Career Highlights
|
Angelic Ventures LP, a family office investing in early-stage technology and data companies • Founder and Managing Partner (2012-present) Thomson Reuters Corporation, multi-national media and information firm • Chief Executive Officer (2008-2011) • Chief Executive Officer, Reuters Group PLC (2001-2008)
|
Other Public Directorships
|
Current • Morgan Stanley (since 2013) • Publicis Groupe (since 2016) Former • None
|
Merck & Co., Inc. 2022 Proxy Statement
|
|
Risa J. Lavizzo-Mourey, M.D. Independent
|
| ||||||||||||
Age:
| ||||||||||||||
Director Since: 2020
| ||||||||||||||
Committees:
| ||||||||||||||
|
| |||||||||||||
Compensation and Management Development
| Research | |||||||||||||
| ||||||||||||||
Experience
|
Dr. Lavizzo-Mourey has extensive health policy experience, serving as Robert Wood Johnson Foundation
|
Career Highlights
|
University of Pennsylvania • Robert Wood Johnson Foundation Population Health and Health Equity Professor Emerita (2021-Current) • Penn Integrates Knowledge Professor of Health Equity and Health Policy Robert Wood Johnson Foundation • President Emerita (2017-present) • President and Chief Executive Officer (2003-2017) • Senior Vice President and Director (2001-2002)
|
Other Public Directorships
|
Current • • Intel Corporation (since 2018) Former • Better Therapeutics
• Hess Corporation (2004-2020)
|
Merck & Co., Inc. 2024 Proxy Statement
Proposal 1 Election of Directors | ÷ ÷ ÷ ÷ | 39 |
|
Stephen L. Mayo, Ph.D. Independent
|
| ||||||||||
Age:
| ||||||||||||
Director Since: 2021
| ||||||||||||
Committees:
| ||||||||||||
 |  | |||||||||||
Audit |
Research | |||||||||||
| ||||||||||||
Experience
|
Dr. Mayo has extensive scientific experience relevant to the biopharmaceutical industry, including being the Bren Professor of Biology and Chemistry, Merkin Institute Professor and former Chair of the Division of Biology and Biological Engineering at the California Institute of Technology (“Caltech”) and co-founder of Xencor, a public antibody engineering company. In addition, in his role as the former Vice Provost at Caltech, Dr. Mayo oversaw Caltech’s technology licensing program. Elected to the National Academy of Sciences in 2004 for his pioneering contributions in the field of protein design, Dr. Mayo has also served as a presidential appointee on the National Science Foundation’s National Science Board and as an elected board member for the American Association for the Advancement of Science. Dr. Mayo also serves as a member of the board of directors of Sarepta Therapeutics, Inc and Allogene Therapeutics, Inc. |
Career Highlights
|
California Institute of Technology • Merkin Institute Professor (2021-present) • Bren Professor of Biology and Chemistry (2007–present) • Chair, Division of Biology and Biological Engineering (2010–2020) • Vice Provost for Research (2007–2010) Howard Hughes Medical Institute, non-profit medical research organization • Investigator (1994–2007)
|
Other Public Directorships
|
Current • Allogene Therapeutics (since 2021) • Sarepta Therapeutics (since 2021) Former • None
|
Merck & Co., Inc. 2022 Proxy Statement
|
Paul B. Rothman, M.D. Independent
|
| ||||||||||
Age:
| ||||||||||||
Director Since: 2015
| ||||||||||||
Committees:
| ||||||||||||
 |
| |||||||||||
Audit | Research (Chair)
| |||||||||||
Experience
|
Dr. Rothman has extensive expertise in patient care, science and medicine relevant to the pharmaceutical industry, including through his |
Career Highlights
|
Johns Hopkins University • Dean of the Medical Faculty and Vice President for Medicine Johns Hopkins Medicine • Chief Executive Officer Carver College of Medicine at the University of Iowa • Dean (2008-2012) • Head of Internal Medicine (2004-2008)
|
Other Public Directorships
|
Current • Former • None
|
Merck & Co., Inc. 2024 Proxy Statement
| 40 | ç ç ç ç | Proposal 1 Election of Directors |
|
Patricia F. Russo Independent
|
| ||||||||||
Age:
| ||||||||||||
Director Since: 1995(1)
| ||||||||||||
Committees:
| ||||||||||||
 |  | |||||||||||
Compensation and Management Development (Chair)
|
Governance | |||||||||||
Experience
|
Ms. Russo has extensive management, operational, international business and financial expertise, as well as a broad understanding of the technology industry, which includes her career achievements during her tenure as CEO and Director of Alcatel-Lucent and Lucent Technologies Inc. In addition, her directorships at other public companies, including her roles as the Non-executive
|
Career Highlights
|
Hewlett Packard Enterprise Company, technology company • Non-executive Alcatel-Lucent, global telecommunications equipment company • Chief Executive Officer and Director (2006-2008) • Chairman, Lucent Technologies Inc. (2003-2006) • President and Chief Executive Officer, Lucent Technologies Inc. (2002-2006)
|
Other Public Directorships
|
Current • General Motors Company (since 2009) • Hewlett Packard Enterprise Company (since 2015), Non-executive • KKR Management Inc. (the managing partner of KKR & Co., L.P.) (since 2011) Former • Arconic, Inc. (2016-2018) formerly Alcoa, Inc. (2008-2016)
|
| (1) | Ms. Russo was on the Board of Directors of Schering-Plough Corporation from 1995 until 2009 when the Company became Merck & Co., Inc. |
Merck & Co., Inc. 2022 Proxy Statement
|
|
Christine E. Seidman, M.D. Independent
|
| ||||||||||
Age:
| ||||||||||||
Director Since: 2020
| ||||||||||||
Committees:
| ||||||||||||
 |  | |||||||||||
Audit |
Research
| |||||||||||
| ||||||||||||
Experience
|
Dr. Seidman has extensive scientific experience relevant to the biopharmaceutical industry, including being the Thomas W. Smith Professor of Medicine and Genetics at Harvard Medical School and the director of the Cardiovascular Genetics Center.
Awards • The Ray C. Fish Award for Scientific Achievement (2020) • American Heart Association Medal for Genomic and Precision Medicine (2019) • Vanderbilt Prize in Biomedical Sciences (2019) |
Career Highlights
|
Harvard Medical School/Brigham and Women’s Hospital (Harvard University) • Thomas W. Smith Professor of Medicine and Genetics (2005-present) • Professor of Genetics and Medicine (1998-2005) • Professor of Medicine (1997-1998) Howard Hughes Medical Institute, non-profit medical research organization • Investigator (1994-present) Brigham and Women’s Hospital • Director, Cardiovascular Genetics Center (1992-present) • Attending Physician, Cardiovascular Division (1987-present)
|
Other Public Directorships
|
Current • None Former • None
|
Merck & Co., Inc. 2024 Proxy Statement
Proposal 1 Election of Directors | ÷ ÷ ÷ ÷ | 41 |
|
Inge G. Thulin Independent
|
| ||||||||
Age:
| ||||||||||
Director Since: 2018
| ||||||||||
Committees:
| ||||||||||
 |  | |||||||||
Compensation and Management Development
|
Governance | |||||||||
| ||||||||||
Experience
|
Mr. Thulin has extensive management, operational, technology and international business expertise, as demonstrated by a track record of success leading 3M Company. Mr. Thulin possesses broad industry experience drawn from 3M’s diverse businesses, commitment to research and strong life sciences division. He also brings valuable insight into driving innovation, based on his experience with new product development and manufacturing. In addition, his previous directorships at other public companies provide him with deep experience on governance issues facing large public companies. |
Career Highlights
|
3M Company, global technology company • Executive Chairman (2018-2019) • Chairman, President and Chief Executive Officer (2012-2018) • President and Chief Executive Officer (2012) • Executive Vice President and Chief Operating Officer (2011-2012) • Executive Vice President, International Operations (2004-2011)
|
Other Public Directorships
|
Current • None Former • 3M Company (2012-2019) • Chevron Corporation (2015-2019)
|
Merck & Co., Inc. 2022 Proxy Statement
|
Kathy J. Warden Independent
|
| ||||||||||
Age:
| ||||||||||||
Director Since: 2020
| ||||||||||||
Committees:
| ||||||||||||
 |  | |||||||||||
Audit |
Governance | |||||||||||
Experience
|
Ms. Warden has broad experience in operational leadership |
Career Highlights
|
Northrop Grumman Corporation,global security company • • President and Chief Operating Officer (2018) • Corporate Vice President and President, Mission System Sector (2016-2017) • Corporate Vice President and President, Information Systems Sector (2013-2015) • Vice President, Cyber Intelligence Division (2011-2012)
|
Other Public Directorships
|
Current • Northrop Grumman Corporation Former • None
|
| ||||||||||
| ||||||||||
| ||||||||||
| ||||||||||
 |  | |||||||||
|
| |||||||||
|
|
|
|
|
|
Merck & Co., Inc. 2022 2024 Proxy Statement
| 42 | ||||||||||
ç ç ç ç |
|
Our non-employee Directors receive cash compensation, as well as cash-settled equity compensation in the form of deferred stock units, for their Board service. During 2021, 2023, non-employee Directors were compensated for their Board service as shown in the chart below.
20212023 Schedule of Director Fees
Compensation Element(1) | Director Compensation Program | |
Annual Retainer | $120,000 | |
Annual Mandatory Deferral | $ the Plan for Deferred Payment of Directors’ Compensation | |
Committee Chair Retainer | $30,000 for the Audit Committee(2) | |
$20,000 for the Governance Committee(3) | ||
$20,000 for the Compensation and Management Development Committee | ||
$20,000 for the Research Committee | ||
Audit Committee Member Retainer | $10,000(2) | |
Lead Director Retainer | $40,000(3) |
| (1) | All compensation is annual. Retainers are paid in quarterly installments and may be voluntarily deferred at the Director’s election. |
| (2) | The Audit Committee Chair retainer includes the Audit Committee Member retainer fee in the amount of $10,000. |
| (3) | The Lead Director is the Chair of the Governance Committee as prescribed by the Governance Committee charter. As a result of the combined responsibility, the Lead Director retainer totals $60,000 in the aggregate. |
Directors’ Deferral Plan
Annual Retainer
Under the Merck & Co., Inc. Plan for Deferred Payment of Directors’ Compensation (“Directors’ Deferral Plan”), each Director may elect to defer all or a portion of cash compensation from retainers. Any amount so deferred is, at the Director’s election, valued as if invested in investment measures offered under the Merck U.S. Savings Plan, including our common stock, and is payable in cash installments or as a lump sum generally no sooner than one year after service as a Director ceases.
Annual Mandatory Deferral
In addition to the annual retainer, upon election (or re-election) at thean Annual Meeting of Shareholders, each Director receives a credit, which for 2021,2023, was valued at $200,000$220,000 in the form of phantom shares denominated in Merck common stock to the Director’s account under the Directors’ Deferral Plan. Directors who join the Board after thean Annual Meeting of Shareholders are credited with a pro-rata portion. All distributions from the Directors’ deferred account are payable in cash installments or as a lump sum and are generally made no sooner than one year after service as a Director ceases.
Expenses and Matching Gift Program
We reimburse all Directors for travel and other necessary business expenses incurred in the performance of their
services for us. We also extend coverage to Directors under our travel accident and directors’ and officers’ indemnity insurance policies. Directors are also eligible to participate in the Merck Foundation Matching Gift Program. The maximum gift total for an active Director participating in the matching gift program is $30,000 in any calendar year.
Director Stock Ownership Guidelines
Upon joining the Board, each Director must own at least one share of Merck common stock. Directors must attain a target Merck common stock ownership level having a value equal to five times the annual cash retainer within five years of joining the Board, or as soon thereafter as practicable. Deferred stock units held in the Merck common stock account under the Directors’ Deferral Plan are counted toward the target goal. Any Director may request that the Governance Committee consider whether the target ownership level is appropriate in view of such Director’s personal circumstances.
As of December 31, 2021,2023, all Directors serving at least three years have either met or exceeded these stock ownership requirements. Dr. Seidman and Ms. Warden joined the Board effective March 16, 2020, Dr. Lavizzo-MoureyMr. Baker joined the Board effective May 26, 202024, 2022 and, Dr. Mayo joinedas of the Board effective March 15, 2021. Each of these Directors is making progress toward meeting thedate hereof, has met his stock ownership guidelines.requirements.
Merck & Co., Inc. 2022 2024 Proxy Statement
2023 Director Compensation
|
÷ ÷ ÷ | 43 |
20212023 Director Compensation
The table below summarizes the annual compensation for our non-employee Directors for the fiscal year ended December 31, 2021.2023.
Mr. Davis and Mr. Frazier areis the only DirectorsDirector who are officerswas an officer and employeesemployee of the Company during 2023, and they dohe did not receive any additional compensation for theirhis Board service.service in 2023.
| Director Compensation for Fiscal Year Ended December 31, 2021 | ||||||||||||
Name | Fees Earned or Paid in Cash ($) | All Other Compensation ($)(3) | Total ($) | |||||||||
Leslie A. Brun(1) |
| $120,815 |
|
| $200,000 |
| $ | 320,815 |
| |||
Thomas R. Cech, Ph.D.(1) |
| 62,500 |
|
| 30,751 |
|
| 93,251 |
| |||
Mary Ellen Coe |
| 130,000 |
|
| 200,000 |
|
| 330,000 |
| |||
Pamela J. Craig |
| 150,000 |
|
| 225,000 |
|
| 375,000 |
| |||
Thomas H. Glocer |
| 150,000 |
|
| 200,000 |
|
| 350,000 |
| |||
Risa J. Lavizzo-Mourey, M.D. |
| 120,000 |
|
| 204,500 |
|
| 324,500 |
| |||
Stephen L. Mayo, Ph.D.(2) |
| 103,441 |
|
| 209,140 | (4) |
| 312,581 |
| |||
Paul B. Rothman, M.D. |
| 141,667 |
|
| 230,000 |
|
| 371,667 |
| |||
Patricia F. Russo |
| 125,000 |
|
| 200,000 |
|
| 325,000 |
| |||
Christine E. Seidman, M.D. |
| 130,000 |
|
| 225,000 |
|
| 355,000 |
| |||
Inge G. Thulin |
| 120,000 |
|
| 200,000 |
|
| 320,000 |
| |||
Kathy J. Warden |
| 130,000 |
|
| 200,000 |
|
| 330,000 |
| |||
Peter C. Wendell |
| 120,000 |
|
| 230,000 |
|
| 350,000 |
| |||
| Director Compensation for Fiscal Year Ended December 31, 2023 | ||||||||||||
Name | Fees Earned or Paid in Cash ($) | All Other Compensation ($)(1) | Total ($) | |||||||||
Douglas M. Baker, Jr. | $127,639 | $220,000 | $347,639 | |||||||||
Mary Ellen Coe | 122,361 | 220,000 | 342,361 | |||||||||
Pamela J. Craig | 150,000 | 250,000 | 400,000 | |||||||||
Thomas H. Glocer | 180,000 | 250,100 | 430,100 | |||||||||
Risa J. Lavizzo-Mourey, M.D. | 120,000 | 245,000 | 365,000 | |||||||||
Stephen L. Mayo, Ph.D. | 130,000 | 220,000 | 350,000 | |||||||||
Paul B. Rothman, M.D. | 150,000 | 230,000 | 380,000 | |||||||||
Patricia F. Russo | 140,000 | 220,000 | 360,000 | |||||||||
Christine E. Seidman, M.D. | 130,000 | 220,000 | 350,000 | |||||||||
Inge G. Thulin | 120,000 | 220,000 | 340,000 | |||||||||
Kathy J. Warden | 130,000 | 220,000 | 350,000 | |||||||||
Peter C. Wendell(2) | 120,000 | 250,000 | 370,000 | |||||||||
| (1) |
|
|
Represents credits in the form of cash-settled deferred stock units (phantom shares) of Merck common stock to the Directors’ Deferral Plan. |
Figures also include charitable contributions made by the Merck Foundation under its matching gift program on behalf of the following Directors: |
Director Name | Matched Charitable Contribution ($) | |||
Cech* | $ | 30,751 |
| |
Craig |
| 25,000 |
| |
Lavizzo-Mourey |
| 4,500 |
| |
Rothman |
| 30,000 |
| |
Seidman |
| 25,000 |
| |
Wendell |
| 30,000 |
| |
*$900 of Dr. Cech’s $30,751 matched charitable contribution was requested in 2020, but not paid until 2021.
Director Name | Matched Charitable Contribution ($) | |||
Craig | 30,000 | |||
Glocer | 30,100 | * | ||
Lavizzo-Mourey | 25,000 | |||
Rothman | 10,000 | |||
Wendell | 30,000 | |||
* Includes $25,000 of charitable contributions made by the Merck Foundation in 2023 in connection with gifts made by Mr. Glocer during the 2022 calendar year. |
| |||
|
Changes to Non-Employee Director Compensation Program effective 20222024
The Governance Committee reviews the Company’s non-employee Director compensation program on a biennial basis. In 2021,2023, the Governance Committee conducted such a review in consultation with FW Cook, the C&MD Committee’s independent compensation consultant.
The review included FW Cook’s analysis of both compensation levels and program design compared to Merck’s peer groups that are used for executive compensation competitive benchmarking—a U.S. pharmaceutical peer group and a supplemental peer group comprised of the Dow Jones Industrial Average companies, excluding financial services companies (as described on page 47)49). Based on this review and the recommendation of FW Cook, the Governance Committee submitted its findings to the full Board in November 20212023 and recommended that the full Board approve changes to the non-employee Director compensation program to address anticipated market trends and increased ratesbetter align leadership position retainers with external peer companies prior to the Governance Committee’s biennial Director compensation program review in 2023.2025. Based on the results of FW Cook’s analysis and the Governance Committee’s recommendation, the Board approved the following changes to the non-employee Director compensation program effective JanuaryApril 1, 2022:2024:
|
The above increase represents a 6% increase in total non-employeeIncreased Lead Director compensation. retainer from $40,000 to $50,000; and
Increased Committee chair retainer from $20,000 to $25,000.
The Governance Committee will continue to conduct, on a biennial basis, a competitive assessment of our non-employee Director compensation program with the goal of maintaining it at or near the median of our external peer groups.
Merck & Co., Inc. 2022 2024 Proxy Statement
|
44 |
| ||||||||||
| | | | | Proposal 2 Non-Binding Advisory Vote to |
We are pleased to provide our shareholders the opportunity to vote on a non-binding, advisory resolution to approve the compensation of our Named Executive Officers as disclosed in this proxy statement, including the Compensation Discussion and Analysis, compensation tables and the narrative discussion accompanying the tables, beginning on page 43.45. As described in the CD&A, our executive compensation programs are principally designed to reward executives based on the achievement of Company and individual performance objectives which, as a whole, are intended to drive sustainable long-term value creation for shareholders and reflect and maintain our position as an industry leader in the development of innovative medicines. The compensation of our NEOs is also designed to enable us to attract, engage and retain talented, high-performing and experienced executives in a competitive market.
In order to align executive pay with operational performance and the creation of long-term shareholder value, a significant portion of compensation paid to our NEOs is allocated to annual cash incentives and long-term equity incentives, which are both directly linked to Company and/or stock price performance. For 2021,2023, approximately 90%91% and 83%82%, respectively, of the CEO’s and other NEOs’ annual target total direct compensation was variable based on our operating performance and/or our stock price.
In addition, management and the C&MD Committee continually review the compensation programs for the NEOs to ensure they achieve the desired goals of reinforcing alignment of officer incentives with the interests of shareholders and linking compensation to performance as measured by operational results. As a result, we have adopted the policies and practices described on page 4648 to further align pay with operational performance and increases in long-term shareholder value while minimizing incentives that could lead to excessive risk-taking.
We are asking shareholders to indicate their support for the NEO compensation as described in this proxy statement. Accordingly, the following resolution will be submitted for approval by shareholders at the 20222024 Annual Meeting:
“Resolved, that the compensation paid to the Company’s Named Executive Officers, as disclosed pursuant to Item 402 of Regulation S-K, including the Compensation Discussion and Analysis, compensation tables and the narrative discussion described in pages 43-8145-87 of this proxy statement, is hereby APPROVED on an advisory basis.”
The shareholder vote on this resolution will not be binding on management, the C&MD Committee or the Board and will not be construed as overruling any decision by management, the C&MD Committee or the Board. However, the Board and the C&MD Committee value the opinions of our shareholders as expressed through their votes and other communications. In 2021,2023, shareholders continued their support of our executive compensation programs with approximately 91% of the votes cast for approval of a similar proposal. We will continue to give careful consideration to the outcome of the advisory vote on executive compensation and to the opinions of our shareholders when making compensation decisions.
At our 20172023 Annual Meeting of Shareholders, our shareholders voted in support of annual advisory votes on future executive compensation proposals. The Board of Directors has adopted a practicepolicy providing for annual say-on-pay“say on pay” advisory votes. The Board expects that the next say-on-pay“say on pay” vote will occur in 2023.2025.
FOR
The Board of Directors recommends that shareholders vote FOR the resolution to approve, on an advisory basis, the compensation of our Named Executive Officers.
|
Merck & Co., Inc. 2022 2024 Proxy Statement
Compensation Discussion and Analysis | | | | | |
45 |
| |||||||
This CD&A describes the material elements of compensation for our 20212023 Named Executive Officers.
Named Executive Officers
Robert M. Davis
Chairman, Chief Executive Officer and |
Executive
|
Executive Vice President and
|
President, |
Executive Vice President and President, Merck |
|
Table of Contents
Executive | |||||||
| 47 | |||||||
| |||||||
| |||||||
| 60 | |||||||
Merck & Co., Inc.2022 2024 Proxy Statement
| 46 | ||||||||||
ç ç ç ç |
Compensation Discussion and Analysis Executive Summary |
2021 was a year of significant achievement and meaningful progress for Merck in the face of what hasIn 2023, our Company continued to be a challenging environment for individuals, healthcare systems,make progress on developing and economiesdelivering transformative therapies and vaccines to help save and improve lives around the world. We remained committed to protecting the health and safety of our employees, sustaining the supply of our medicines and vaccines, employing our scientific capabilities in the global fight against COVID-19 and progressing our pipeline. We achieved strong operational performance acceleratedand made significant advancements in our broad pipeline, completedincluding with strategic acquisitions, collaborations, and partnerships to provide patients the spin-offnext generation of Organon,innovations.
The excellence of our commercial and completed key strategic business development transactions. Despite challenges from the ongoing pandemic, our teams performed with agility and executed with excellence.
Our efforts and underlying business strengthoperational execution enabled us to deliver 17%12% sales growth (16% excluding(excluding the impact of currency exchange)foreign exchange and sales of LAGEVRIO). As a result, we exceeded ourthe Revenue and Pre-Tax Income targets for our 2021 Scorecard. Commercially, we executed extremely well across all our key performance drivers, including KEYTRUDA, GARDASIL / GARDASIL 9, and Animal Health.
KEYTRUDA grew 18% (ex-exchange) to over $17 billion, reflecting continued robust global demand. In the U.S., KEYTRUDA continued to demonstrate durable momentum across all key tumors, and benefited from the recent launches in triple-negative breast cancer (neoadjuvant/adjuvant), renal cell carcinoma (adjuvant and advanced settings), advanced cervical cancer, and advanced endometrial cancer. Outside the U.S., KEYTRUDA growth continued to be driven by non-small cell lung cancer indications and the ongoing launches in head and neck and RCC (adjuvant and advanced settings). In addition, we continued to fortify our position in oncology with several milestones highlighting our strategy of targeting earlier-stage cancers where there is potential for improved outcomes by reducing the risk of recurrence. GARDASIL / GARDASIL 9 grew by 39% (ex-exchange) driving strong growth in our vaccines portfolio. Outside the U.S., robust growth2023 Scorecard. Our commercial success was driven by robust performance across key areas, particularly Oncology, Vaccines, and Animal Health. KEYTRUDA experienced exceptional growth of 21% (excluding foreign exchange), reaching over $25 billion in sales, driven by increased uptake in earlier-stage cancers and continued strong underlying demand across all key geographies, particularly China. GARDASIL / global need of patients with metastatic disease. Our vaccines portfolio also showed strong growth, with GARDASIL/GARDASIL 9 are increasingly being recognized as vaccines that can help prevent certain HPV-related cancerssales growing by 33% (excluding foreign exchange), due to strong global demand, particularly in both females and males.China. Our continued launch of VAXNEUVANCE, for the prevention of pediatric pneumococcal disease, generated $665 million in sales. Our Animal Health business crossedachieved solid growth of 3% (excluding foreign exchange), driven equally by Companion Animal and Livestock product segments.
In addition to commercial success, our Company made significant progress in our pipeline. We initiated over 20 phase 3 studies across eight novel assets and received over 25 regulatory approvals in major markets around the $5.5 billion revenue threshold with 16% growth (ex-exchange)world, highlighting the breadth and is well-positioneddepth of our pipeline. Examples of our progress in 2023 include the FDA’s grant of priority review for continued success.
We also continued to progress onsotatercept, our pipeline, receiving more than 30 approvals and filing more than 20 new drug applications and supplemental biologics license applications inCompany’s novel investigational activin signaling inhibitor, for the U.S., European Union, Japan, and China. In particular, we made meaningful advancements across our broader pipeline with approvals for new molecular entities, including VAXNEUVANCE, Verquvo and WELIREG. For molnupiravir, our investigational oral antiviral COVID-19 medicine, we have received authorizations in 10 countries, including an emergency use authorization (“EUA”) in the U.S. and a special approval for emergency use in Japan. We believe molnupiravir will be an important treatment option to combat the ongoing COVID-19 pandemic with clinical trial data showing significantly reduced hospitalization or death inof adult patients at high risk of progressing to severe disease. Followingwith pulmonary arterial hypertension, the FDA’s EUAgrant of priority review for our new Biologics License Application for V116, our Company’s investigational 21-valent pneumococcal conjugate vaccine specifically designed to help prevent invasive pneumococcal disease and pneumococcal pneumonia in December 2021, our dedicated teams worked diligently to deliver 1.4 million coursesadults, and the FDA’s approval of therapy toan additional indication for WELIREG in patients with advanced renal cell carcinoma, making it the U.S., Japan, United Kingdom, and other countries.first novel therapeutic class available for this population since 2015.
We also acted on keyOur pipeline advancements were complemented by strategic business development opportunities to augment our pipeline,activities, including the acquisitions of Pandion Therapeutics,Prometheus Biosciences, Inc. and Imago BioSciences, Inc., and a clinical-stage biotechnology company developing novel therapeutics designedcollaboration with Daiichi Sankyo. These initiatives expanded our Company’s capabilities and potential reach in the fields of autoimmune conditions, hematology, and antibody-drug conjugates across multiple types of cancer.
Furthermore, we are proud of our ability to addresshave reached more than 500 million people in 2023 with our medicines and vaccines. Our science-led strategy, which keeps patients at the unmet needscenter of patients living with autoimmune diseases,everything we do, helps drive long-term value creation. As previewed in last year’s proxy and Acceleron Pharma Inc., a biopharmaceutical companysummarized below, we increased our focus on driving sustainable business outcomes by linking the compensation of most employees, including our executives, to sustainability metrics focused on harnessingdriving greater access to health to patients around the powerworld and on the engagement and inclusion of the transformingemployees.
In summary, our success in 2023 was driven by exceptional operational execution, robust commercial growth, factor (TGF)-beta superfamily of proteins, with a lead candidate, sotatercept, having the potentialsignificant advancements in our pipeline, and strategic business development. These achievements position our Company for continued success in discovering and delivering innovative therapies and vaccines to provide a novel approach to treating pulmonary arterial hypertension.
Lastly, 2021 marked significant changes within the Merck executive team, including the transition of Kenneth C. Frazier to Executive Chairmansave and Robert M. Davis to Chief Executive Officer, both effective July 1, 2021. The Board’s succession planning resulted in the election of Dean Y. Li, M.D., Ph.D. to succeed Roger M. Perlmutter, M.D., Ph.D. as Executive Vice President and President, Merck Research Laboratories, effective January 1, 2021, and Caroline Litchfield to succeed Mr. Davis as Chief Financial Officer, effective April 1, 2021.improve lives.
Scorecard Performance |
Financial Performance(2) | ||||||||||||||||||||
Target($B) | Actual($B) | Weighting% | Score% | |||||||||||||||||
Revenue |
| $52.80 |
|
| $54.87 |
|
| 40 | % |
| 173 | % | ||||||||
Pre-Tax Income |
| $19.72 |
|
| $20.49 |
|
| 40 | % |
| 132 | % | ||||||||
Non-Financial Performance | ||||||||||||||||||||
Pipeline |
| 20 | % |
| 130 | % | ||||||||||||||
Overall Payout |
|
| 148 | % | ||||||||||||||||
Financial Performance | ||||||||||||||||||||
Target($B) | Actual($B) | Weighting | Score | |||||||||||||||||
Revenue |
| $57.94 |
|
| $60.29 |
|
| 35 | % |
| 158 | % | ||||||||
Pre-Tax Income |
| $21.20 |
|
| $22.52 |
|
| 35 | % |
| 152 | % | ||||||||
Non-Financial Performance | ||||||||||||||||||||
Pipeline |
| 20 | % |
| 147 | % | ||||||||||||||
Sustainability |
| 10 | % |
| 100 | % | ||||||||||||||
Total Payout |
|
| 148 | % | ||||||||||||||||
PSU Performance |
| Peer Median | Merck | Result | Weighting | Payout | ||||||||||||||||||||
3-Year R-TSR |
| 9.86% |
|
| 5.90% |
|
| 80% |
|
| 50% |
|
| 40% |
| |||||||||
| Target($B) | Actual($B) | Result | Weighting | Payout | ||||||||||||||||||||
2-Year Cum. OCF |
| $28.80 |
|
| $32.60 |
|
| 200% |
|
| 25% |
|
| 50% |
| |||||||||
2-Year Cum. EPS |
| $9.64 |
|
| $10.97 |
|
| 200% |
|
| 25% |
|
| 50% |
| |||||||||
Overall Payout |
|
| 100% |
|
| 140% |
| |||||||||||||||||
| Target | Actual | Weighting | Score | |||||||||||||||||
1-Year EPS(2) |
| $6.58 |
|
| $7.17 |
|
| 33% |
|
| 175% |
| ||||||||
| Peer Median | Merck | Weighting | Score | |||||||||||||||||
3-Year R-TSR(2) |
| 3.70% |
|
| 14.60% |
|
| 67% |
|
| 154% |
| ||||||||
Total Payout |
|
| 161% |
| ||||||||||||||||
| (1) |
|
| (2) |
|
Merck & Co., Inc. 2022 2024 Proxy Statement
Compensation Discussion and Analysis Executive Compensation Overview |
÷ ÷ ÷ | 47 |
Executive Compensation Program Objectives and StrategyOverview
The pharmaceutical industry is science-focused and requires experimentation and long-term commitment of financial resources to foster innovation. The Company is at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. Ultimately, our work has the potential to have an enormous impact on global health and well-being. Because of the inherent complexity and dynamic science of human and animal health, even with flawless execution, we risk failure. In addition:
The costs associated with innovation are increasing while relative return is decreasing due to ongoing pricing pressure.
The number of products available to treat or prevent a particular disease or condition typically increases over time, which can limit the commercial potential of key products.
It generallyGenerally, it takes 10 to 15 years to discover, develop, and bring a new productmedicine or vaccine to market.
Competition for qualified talent in the pharmaceutical industry, both in the U.S. and Pay-for-Performanceinternationally, is intense.
Our Executive Compensation Strategy
We strive to balance the need to deliver market-competitive pay within a framework that provides the appropriate mix of fixed and variable, at-risk compensation to attract, retain, and motivate talent and align with our pay-for-performance objectives.
Our executive compensation program is designed to… | |||||
 | Support our efforts to attract and retain the brightest and most innovative minds in business, research, and academia. | ||||
| Align the interests of our executives with the interests of our shareholders to ensure prudent actions that will | ||||
 | Reward our executives based on the achievement of sustained financial and operational performance and demonstrated leadership. | ||||
| Support a shared, one-company mindset of performance and accountability to deliver on business objectives.
| ||||
Variable Compensation is Critical to Achieve Our Objectives
Annual Cash Incentive
The Company Scorecard (described in more detail on page 53) focuses on our most critical business drivers — Revenue, Pre-Tax Income, and Pipeline accomplishments — and is used to determine the payout for our annual incentive for all employees, including our NEOs under the Executive Incentive Plan. Our Scorecard performance for 2021 resulted in above-target achievement of 148%.
Long-Term Incentive
The long-term incentive program, consisting of a mix of PSUs and stock options, provides our NEOs with the opportunity to own Merck stock, directly linking a substantial portion of their compensation to the returns realized by our shareholders.
The 2019 PSU program (described in more detail on page 57) paid out at 140% based on achievement of cumulative two-year OCF, cumulative two-year EPS and three-year R-TSR metrics during the performance period, weighted at 25%, 25% and 50%, respectively. As previously disclosed, cumulative two-year OCF and EPS metrics were used due to the complexities associated with disentangling our Organon business from a multi-year financial plan. Organon was successfully spun off in June 2021.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 48 | ||||||||||
ç ç ç ç |
Compensation Discussion and Analysis Executive Compensation Overview |

In 2021, shareholders continued their support for our executive compensationprograms with approximately 91% of the votes cast in favor of the say-on-payproposal. Consistent with the Company’s strong interest in shareholderengagement and our pay-for-performance approach, the C&MD Committeecontinues to evaluate our executive compensation program to ensure alignmentbetween the respective interests of our executives and shareholders. The C&MDCommittee did not make significant changes to our executive compensationprogram in 2021 as a direct result of the most recent say-on-pay vote.
We ask that our shareholders approve, on an advisory basis, the compensation ofour NEOs as further described in Proposal 2 on page 42.
Compensation Policies and Practices
Our executive compensation and corporate governance programs are designed to closely link pay with operational performance and increases in long-term shareholder value while minimizing incentives that could lead to excessive risk-taking. To help us accomplish these important objectives, we have adopted the following policies and practices over time:practices:
We do… | We do not… | ||||||
| Utilize a relative total shareholder return metric in the PSU program to align the payout with long-term stock performance and shareholder experience
|
| |||||
| Monitor LTI program share utilization regularly relative to both industry standards and versus our primary and supplemental peer groups | ||||||
| Conduct annual competitive benchmarking to ensure executive officer compensation is aligned to market | ||||||
| Include caps on annual cash incentive and PSU program payouts and thresholds below which no payouts are earned | ||||||
| Offer limited perquisites that are supported by business interests | ||||||
| Retain an independent compensation consultant that reports directly to the C&MD Committee | ||||||
| Maintain robust stock ownership requirements and share retention policies | ||||||
| Maintain a rigorous incentive recoupment (i.e., clawback) policy that exceeds the NYSE listing requirements | ||||||
| Conduct assessments to identify and mitigate risk in our compensation programs | ||||||
|
Provide dividend equivalents only on earned Restricted Stock Units (“RSUs”) and PSUs
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
Require double-trigger vesting of equity in the event of a change in control (i.e., there must be both a change in control and an involuntary termination)
| ||||||
|
Avoid employment agreements
| ||||||
Say-on-Pay Advisory Vote In 2023, shareholders continued their historically strong support for our executive compensation programs with approximately 91% of the votes cast in favor of the say-on-pay proposal. Based on this outcome and the C&MD Committee’s ongoing analysis of the program’s ability to support our strategic, financial, and human capital objectives, we did not make significant changes to our executive compensation program in 2023. Consistent with the Company’s strong interest in shareholder engagement and our pay-for-performance approach, the C&MD Committee continues to evaluate our executive compensation program to ensure alignment between the respective interests of our executives and shareholders. |  |
We ask that our shareholders approve, on an advisory basis, the compensation of our NEOs as further described in Proposal 2 on page 44.
Merck & Co., Inc. 2022 2024 Proxy Statement
Compensation Discussion and Analysis Peer Groups |
÷ ÷ ÷ | 49 |
Individual executive officer compensation levels and opportunities are compared to a peer group of large multinational pharmaceutical companies approved by the C&MD Committee that participate in a pharmaceutical industry compensation survey conducted by Willis Towers Watson, an independent consulting firm. In setting compensation levels for 2021,2023, the C&MD Committee reviewed the survey results for the following peer companies that Merck competes with to attract talented, high-performing executives.executives (the “primary peer group”). The C&MD Committee occasionallyregularly reviews segmented information related tofocused on the companies in the primary peer group that are headquartered in the U.S. because practices outside the U.S. can differ geographically.
Primary Peer Group Companies
AbbVie Amgen AstraZeneca Bristol-Myers Squibb Eli Lilly Gilead Sciences GlaxoSmithKline Johnson & Johnson Novartis Pfizer Roche Holding AG Sanofi
All numbers are as of 12/31/
|
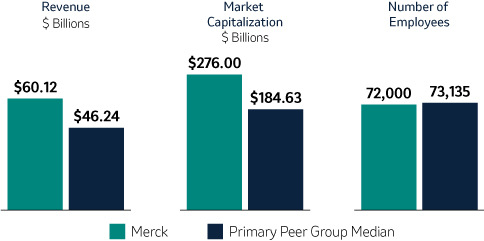 |
In 2021, the C&MD Committee approved the addition of Gilead Sciences, Inc. to our primary peer group, effective January 1, 2022.
Merck’s Supplemental Peer Group
In addition to the pharmaceuticalprimary peer group described above, we also use a supplemental peer group consisting of the companies that comprise the Dow Jones Industrial Average (excluding the financial services companies) as a secondary reference for CEO compensation and for other compensation-related practices (for example, share usage and dilution, change in control policy design, and stock ownership and retention guidelines). Merck is a member of the Dow Jones Industrial Average, and we believe this group provides insight into practices among companies of similar scale and complexity that operate across a variety of industries, providing us with a broader view of market pay, policies, and practices.
Supplemental Peer Group Companies(1)
|
| |||
3M Amgen Apple Boeing Caterpillar Chevron Cisco Systems Coca-Cola Dow Home Depot Honeywell IBM Intel | Johnson & Johnson McDonald’s Microsoft Nike Procter & Gamble salesforce.com UnitedHealth Group Verizon Visa Walgreens Walmart Walt Disney | |||
(1) Reflects Dow Jones Industrial Average companies (excluding the financial services companies) as of the beginning of
All numbers are as of 12/31/
| ||||
Our overarching strategy is to position our executives’ target TDC at the median, on average, with variability by individual executive based on scope and complexity of role, market availability of proven talent, experience, leadership, sustained performance over time, potential for advancement as part of succession planning, and other unique factors that may exist from time to time. This median target compensation philosophy ensures that actual realized compensation varies above or below market levels based on attainment of longer-term goals and changes in shareholder value, and overall costs and share dilution are reasonable and sustainable relative to market practices.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 50 | ||||||||||
ç ç ç ç |
Compensation Discussion and Analysis Detailed Discussion and Analysis |
Detailed Discussion and Analysis
FurtherAdditional information regarding our 20212023 Named Executive Officers and the material elements of their compensation, as reported in the Summary Compensation Table on page 63, is described below.below*.
|
|
Compensation Decisions for
| ||||||||
Robert M. Davis Chairman, Chief Executive Officer
|
• Increased base salary by • • Increased LTI target by • Changes resulted in |
| ||||||||
Age:
| ||||||||||
Tenure*:
| ||||||||||
| ||||||||||
|
Compensation Decisions for | |||||||||
|
|
| ||||||||
| ||||||||||
| ||||||||||
| ||||||||||
Merck & Co., Inc. 2022 Proxy Statement
|
|
| ||||||||
Caroline Litchfield Executive Vice President and Chief Financial Officer | • Increased base salary by $150,000 • Maintained annual incentive target percentage • Increased LTI target by $1,500,000 • Changes resulted in a 38.3% increase in target TDC |
| ||||||
Age: 55 | ||||||||
Tenure*: 33 Years | ||||||||
* Compensation shown for each executive is rounded to the nearest dollar and length of tenure is rounded to the nearest year.
Merck & Co., Inc. 2024 Proxy Statement
Compensation Discussion and Analysis Detailed Discussion and Analysis | ÷ ÷ ÷ ÷ | 51 |
Compensation Decisions for 2023 | ||||||||
Sanat Chattopadhyay Executive Vice President and President, Merck Manufacturing Division
|
• Set base salary at
|
| ||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
|
| ||||||
| ||||||||
| ||||||||
| ||||||||
Merck & Co., Inc. 2022 Proxy Statement
|
| ||||||||
|
• Set annual incentive target at 100% • Set LTI target at
|
| ||||||
Age:
| ||||||||
Tenure*:
| ||||||||
| ||||||||
|
|
Compensation Decisions for | ||||||||
Richard R. DeLuca, Jr. Executive Vice President and President, Merck Animal Health | • Increased base salary by $75,000 • Maintained annual incentive target percentage • Increased LTI target by $200,000 • Changes resulted in a 7.4% increase in target TDC |
| ||||||||
Age: 61 | ||||||||||
Tenure*: 12 Years | ||||||||||
Merck & Co., Inc. 2024 Proxy Statement
| 52 | ç ç ç ç | Compensation Discussion and Analysis Detailed Discussion and Analysis |
Compensation Decisions for 2023
| ||||||||
Dean Li, M.D., Ph.D. Executive Vice President and President, Merck Research Laboratories
|
• • • • Changes resulted in a 31.3% increase in target TDC |
| ||||||
Age:
| ||||||||
Tenure*:
| ||||||||
| ||||||||
Merck & Co., Inc. 2022 2024 Proxy Statement
Compensation Discussion and Analysis The Elements of |
÷ ÷ ÷ | 53 |
The Elements of 20212023 Compensation
How Our Compensation Program Works
What We Reward | How We Link Pay To Performance | How We Pay | ||||||||||||||||
• Top and bottom-line performance that meets or exceeds
• Pipeline accomplishments that advance our position as an industry-leading biopharmaceutical company
• Achievement of strategic sustainability priorities that focus on greater access to health and on the engagement and inclusion of employees • Decision-making that yields long-term value creation for shareholders
•
| • Inclusion of key financial and non-financial metrics in our annual cash incentive plan to ensure executives are rewarded for top and bottom-line performance,
• Long-term
• Majority of total target pay opportunity is at-risk and tied to company performance and/or long-term stock value
| • Overall target total pay opportunity, as well as each pay element, is assessed for competitiveness relative to primary and/or supplemental peer groups, which include
• Competitive positioning is targeted to median of market; actual positioning varies based on a variety of factors, including scope and complexity of role, years of experience, demonstrated performance over time, and other factors
| ||||||||||||||||
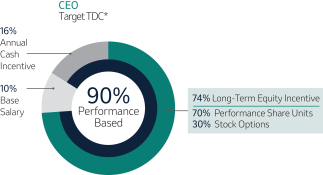
| 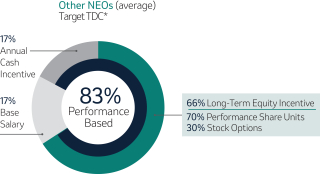 | 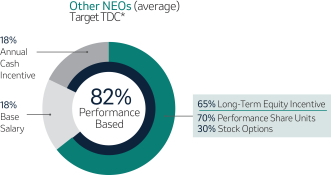 |
*Rounded based on full year long-term incentive and annual incentive targets.targets (excluding one-time special awards).
TheEach year, the C&MD Committee recommends, and the independent members of the Board of Directors approve, the compensation for our CEO and Executive Chairman.CEO. The C&MD Committee reviews and approves compensation for all other NEOs each year based on a variety of factors, including scope and complexity of role, experience, sustained leadership, and performance and competitive positioning as compared to our pharmaceuticalprimary and supplemental peer groups as described in more detail on page 47.49.
Additional details regarding the roles and responsibilities of the C&MD Committee are provided on page 15.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 54 | ||||||||||
ç ç ç ç |
Compensation Discussion and Analysis The Elements of |
The C&MD Committee must balance
The table shows adjustments made to | Named Executive Officer | Annual Base Salary Increase % | Market/Promotional Adjustment % | New Base Salary | ||||||||||||
Davis(1) |
| 3.0 | % |
| 30.7 | % | $ | 1,500,000 |
| |||||||
Frazier(2) |
| NA |
|
| -26.5 |
|
| 1,250,000 |
| |||||||
Litchfield |
| — | (3) |
| — | (3) |
| 900,000 |
| |||||||
Clyburn |
| — | (3) |
| — | (3) |
| 1,000,000 |
| |||||||
DeLuca |
| — | (3) |
| — | (3) |
| 800,000 |
| |||||||
Li |
| — | (3) |
| — | (3) |
| 950,000 |
| |||||||
(1) Annual salary increase, effective March 2021; promotional adjustment effective July 2021. (2) Reduction in base salary effective July 2021. (3) Ms. Litchfield, Mr. Clyburn, Mr. DeLuca and Dr. Li were not NEOs in 2020. |
| |||||||||||||||
The C&MD Committee must balance the need to deliver a competitive level of base salary with ensuring the appropriate mix of fixed to variable compensation for each NEO.
The table shows adjustments made to base salaries in 2023. All annual base salary increases were based on Merck’s U.S. salary increase budget for all employees, including the NEOs. | Named Executive Officer | | Annual Base Salary Increase % |
| | Market Adjustment % | | New Base Salary(1) | ||||||||||
Davis |
| 4.5 | % |
| No change |
|
| $1,615,000 |
| |||||||||
Litchfield |
| 4.5 |
|
| 10.9 | % |
| 1,125,000 |
| |||||||||
Chattopadhyay |
| — | (2) |
| — | (2) |
| 941,806 |
| |||||||||
DeLuca |
| 4.5 | (2) |
| 4.3 | (2) |
| 925,000 |
| |||||||||
Li |
| 4.5 |
|
| 7.5 |
|
| 1,400,000 |
| |||||||||
(1) Reflects base salary as of December 31, 2023. (2) Mr. Chattopadhyay was not an NEO in 2022. Although Mr. DeLuca was not an NEO in 2022, he was an NEO in 2021. As such, pursuant to SEC rules, we have included his 2022 compensation information in the Summary Compensation table.
|
| |||||||||||||||||
Annual Cash Incentive
| ||||||||||||||||||
The NEOs participate in the Executive Incentive Plan (“EIP”).
Award amounts under the EIP are determined based upon achievement of Company performance measures as reflected by the Company Scorecard. The overall EIP award fund cannot exceed 200% of the aggregate total target incentive amount for all participants. The maximum award amount for each NEO for 2023, excluding the impact of the Company Scorecard, is listed in the Grants of Plan-Based Awards table on page 73. | Named Executive Officer | | 2022 Target Annual % of Base Salary(1) |
| | 2023 Target Annual % of Base Salary(1) |
| |||||||||||
Davis |
| 150 | % |
| 150 | % | ||||||||||||
Litchfield |
| 100 |
|
| 100 |
| ||||||||||||
Chattopadhyay |
| — | (2) |
| 100 |
| ||||||||||||
DeLuca |
| 100 | (2) |
| 100 |
| ||||||||||||
Li |
| 100 |
|
| 100 |
| ||||||||||||
(1) Reflects annual incentive targets as of December 31 of the applicable year. (2) Mr. Chattopadhyay was not an NEO in 2022. Although Mr. DeLuca was not an NEO in 2022, he was an NEO in 2021. As such, pursuant to SEC rules, we have included his 2022 compensation information in the Summary Compensation table.
|
| |||||||||||||||||
The NEOs participate in the Executive
Award amounts under the EIP are | Named Executive Officer | 2020 Target Annual % of Base Salary | 2021 Target Annual % of Base Salary(1) | |||||||||
Davis |
| 105 | % |
| 150 | % | ||||||
Frazier |
| 150 |
|
| 100 |
| ||||||
Litchfield |
| — | (2) |
| 100 |
| ||||||
Clyburn |
| — | (2) |
| 100 |
| ||||||
DeLuca |
| — | (2) |
| 100 |
| ||||||
Li |
| — | (2) |
| 100 |
| ||||||
(1) Reflects annual incentive targets as of December 31, 2021. (2) Ms. Litchfield, Mr. Clyburn, Mr. DeLuca and Dr. Li were not NEOs in 2020.
|
| |||||||||||
Merck & Co., Inc. 2022 2024 Proxy Statement
Compensation Discussion and Analysis The Elements of |
÷ ÷ ÷ | 55 |
2021 Merck2023 Company Scorecard
Our Company Scorecard helps translate our strategic priorities into operational terms that enable tracking and measurement of our progress and performance against annual operating goals and critically important long-term strategic drivers of sustainablelong-term value creation, including goals tied to our research and development pipeline — each of which is measured in the context of compliance, health, safety, and environmental outcomes. The Company Scorecard may be adjusted based on an evaluation of these outcomes, recognizing the importance they play in driving Merck’s values and a culture of integrity. For 2021,2023, no such adjustment was applied. We believe creating greater accountability for the sustainable delivery of business goals will help drive financial results and long-term shareholder value. As a result, the C&MD Committee approved a new measure in our 2023 Company Scorecard that links the compensation of most employees, including our executives, to certain Sustainability metrics. The new design reflects our focus on driving greater access to health to patients around the world and on the engagement and inclusion of our employees, each of which is a strategic priority. Revenue and Pre-Tax Income are equally weighted at 35% (previously 40%), each based on the C&MD Committee’s belief that they are the key financial measures of our success during the year. The Pipeline goals are collectively weighted at 20% and are designed to ensure that we are focused on internal and external early discovery opportunities, late-stage clinical development progression, and regulatory filings and approvals. Finally, our new Sustainability metrics are collectively weighted at 10%.
The target, threshold, and stretchmaximum Revenue and Pre-Tax Income goals are set in relation to the Board-approved annual operating plan and the expectations of management. Each year, the Pipeline goals are recommended by the headPresident of Merck Research Laboratories, reviewed by the Research Committee, and approved by the C&MD Committee. The Sustainability metrics are recommended by Merck’s Global Market Access, ESG, and Human Resources teams and approved by the C&MD Committee. Failure to achieve threshold performance on any of the metrics would result in forfeiture of the entire opportunity for that metric. If the combined results of the threefour metrics do not total at least 50, there would be no payout. The overall results of the Company Scorecard are calibrated so individuals may receive between 50% and 200% of their target award opportunity established for the annual performance period. Adjustments are applied to Revenue and Pre-Tax Income results using a consistent framework of adjustments to our reported financial results for incentive program purposes approved by the C&MD Committee to accurately reflect the operating performance of our business. For further explanation of these adjustments, please refer to Appendix B on page 99.106. The 2023 Company Scorecard results are summarized below.
20212023 Company Scorecard(1)
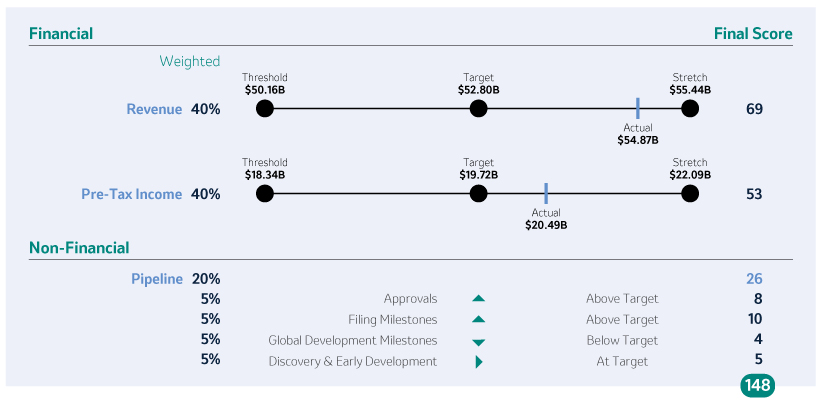
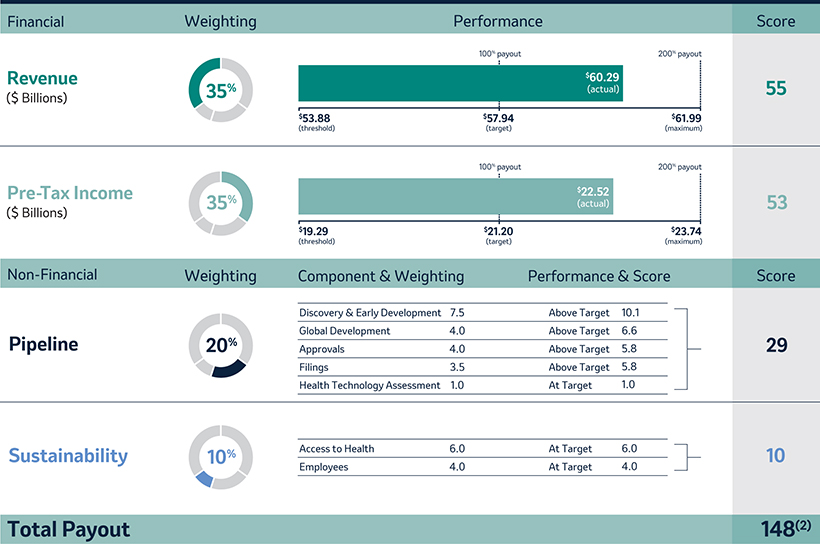
| (1) | Excluding the impact of variances in currency exchange rates versus budget and certain other items, consistent with plan |
| (2) | Rounded to the nearest whole number. |
Revenue:
Merck’s
Merck & Co., Inc. 2024 Proxy Statement
| 56 | ç ç ç ç | Compensation Discussion and Analysis The Elements of 2023 Compensation |
Revenue:
Reported revenue for 2021 was $48.70B, reflecting continuing operations. For purposes of the 2021 Company Scorecard, our internal Revenue goals assumed Organon remained part of Merck for all of 2021. As such, for purposes of determining Revenue for the 2021 Company Scorecard, this figure$60.12B was adjusted to include Organon actual performance prior to spin date and operating plan numbers post spin date. This result of $54.88B was adjusted to $54.87B$60.29B to exclude the impact of currency exchange rates (versus currency exchange rates budgeted in the annual operating plan) and the associated impact of hyper-inflation in certain markets (consistent with plan design).
Pre-Tax Income:
Non-GAAP pre-tax income of $5.99B was adjusted to $22.52B to exclude the effect of certain business development transactions (consistent with plan design and past practice). We exceeded our internal Revenue target of $52.80B due to strong performance in key pillars including oncology, vaccines, and animal health, as well as the significant contribution of molnupiravir.
Merck & Co., Inc. 2022 Proxy Statement
|
Pre-Tax Income:
For purposes of the 2021 Company Scorecard, our internal Pre-Tax Income goals assumed Organon remained part of Merck for all of 2021. As such, Pre-Tax Income from continuing operations was adjusted to include the impact of Organon, similar to the adjustment described above for Revenue. This result of $20.48B was adjusted to $20.49B to exclude the impact of currency exchange rates (versus currency exchange rates budgeted in the annual operating plan) and the effectassociated impact of hyper-inflation in certain business development transactions (consistentmarkets (all consistent with plan design and past practice)design). We exceeded our internal Pre-Tax income target of $19.72B due to the sales strength that was achieved coupled with our continued discipline in expense management.
2021Named Executive Officer 2023 Annual Incentive Payouts
The table below shows the 20212023 annual cash incentives paid to the NEOs. The “Final Award” for each NEO is reflected in the “Non-Equity Incentive Plan Compensation” column of the Summary Compensation table.

Named Executive Officer 2021 Annual Incentive Payments
| Target | ||||||||||||||||||||
Named Executive Officer | Annual Base Salary (as of 12/31/23) ($) | Annual Incentive (%) | Annual Incentive ($) | Company Scorecard Result (%) | Final Award ($) | |||||||||||||||
Davis | $1,615,000 | 150 | % | $2,422,500 | 148 | % | $3,585,300 | |||||||||||||
Litchfield | 1,125,000 | 100 | 1,125,000 | 148 | 1,665,000 | |||||||||||||||
Chattopadhyay | 941,806 | 100 | 941,806 | 148 | 1,393,873 | |||||||||||||||
DeLuca | 925,000 | 100 | 925,000 | 148 | 1,369,000 | |||||||||||||||
Li | 1,400,000 | 100 | 1,400,000 | 148 | 2,072,000 | |||||||||||||||
| Target | ||||||||||||||||||||
Named Executive Officer | Annual Base Salary (as of 12/31/21) ($) | Annual Incentive (%) | Annual Incentive ($) | Company Scorecard Result (%) | Final Award ($) | |||||||||||||||
Davis(1) |
| $1,500,000 |
|
| 150 | % |
| $1,915,274 |
|
| 148 | % |
| $2,834,606 |
| |||||
Frazier(2) |
| 1,250,000 |
|
| 100 |
|
| 1,894,658 |
|
| 148 |
|
| 2,804,094 |
| |||||
Litchfield(3) |
| 900,000 |
|
| 100 |
|
| 800,137 |
|
| 148 |
|
| 1,184,203 |
| |||||
Clyburn(3) |
| 1,000,000 |
|
| 100 |
|
| 975,342 |
|
| 148 |
|
| 1,443,506 |
| |||||
DeLuca |
| 800,000 |
|
| 100 |
|
| 800,000 |
|
| 148 |
|
| 1,184,000 |
| |||||
Li(4) |
| 950,000 |
|
| 100 |
|
| 946,486 |
|
| 148 |
|
| 1,400,799 |
| |||||
|
|
|
|
2021 Equity Award Mix
The long-term incentive program, consisting of a mix of PSUs and stock options, provides our NEOs with the opportunity to own Merck common stock, directly linking a substantial portion of their compensation to the returns realized by our shareholders. We use these two long-term incentive vehicles to ensure that our LTI program remains balanced, sustainable, and supportive of its objectives over a multi-year period.
2023 Equity Award Mix
 | Performance Share Units | |
| PSUs link realized compensation value to the achievement of critical financial | ||
| Stock Options Stock options align our executives’ interests with the interests of our shareholders because options only have financial value to the recipient if the price of our common stock at the time of exercise exceeds the stock price on the date of grant. As a result, we believe stock option grants encourage executives to focus on behaviors and initiatives that support sustained long-term stock price appreciation, which benefits all shareholders. | |
Merck & Co., Inc. 2022 Proxy Statement
|
|
Current LTI Grant Practices
All grants to executive officers are made under the Merck & Co., Inc. 2019 Stock Incentive Plan and approved by the C&MD Committee and, in the case of our CEO, and Executive Chairman, recommended by the C&MD Committee and approved by the independent members of the Board of Directors. Annual PSU grants (with a 3-yearthree-year performance period) are generally made on the last business day in March and annual stock option grants are made on the third business day following the announcement of
Merck & Co., Inc. 2024 Proxy Statement
Compensation Discussion and Analysis The Elements of 2023 Compensation | ÷ ÷ ÷ ÷ | 57 |
our first quarter earnings. We may also selectively grant PSUs, stock options, and RSUs to executive officers on the third business day following the announcement of quarterly earnings generally as part of a new hire sign-on or for retention purposes. These dates were chosen to ensure that grants are made shortly after we have released information about our financial performance to the public. However, the C&MD Committee reserves the right to change the date when grants are made, in view of its responsibility to consider all facts and circumstances to ensure that grants are consistent with our compensation philosophy and objectives.
Stock options are granted at no less than fair market value on a fixed date or date of a particular event, with all required approvals obtained in advance of or on the actual grant date. Fair market value is the closing price of a share of CompanyMerck common stock on the grant date. In certain countries, a higher grant price may be used to satisfy provisions of local applicable law. The re-pricing of stock options is not permitted under the Incentive Stock Plan without prior shareholder approval.
20212023 LTI Grant Values
The 20212023 annual LTI grant values for the CEO and Executive Chairmanother NEOs, as compared to the prior year, are shown in the following table. The number of shares associated with each award is set forth in the Grants of Plan-Based Awards table on page 66.73. The LTI grant value for Mr. Davis was increased by the Board in consideration of his new role as President and Chief Executive Officer. Thethe LTI valuegrant values for Mr. Frazier was decreased by the Board in consideration of his transition to Executive Chairman. The other NEOs were not named executive officersincreased by the C&MD Committee to strengthen their competitive position versus our primary peer group and reflect their expected future contributions in 2020.creating sustained long-term shareholder value.
| Target Grant Value(1) | Increase in Target Grant Value | |||||||||||
Named Executive Officer | 2020 | 2021 | ||||||||||
Davis(2) |
| $4,000,000 |
|
| $9,200,000 |
|
| +$5,200,000 |
| |||
Frazier |
| 15,750,000 |
|
| 10,750,000 |
|
| -5,000,000 |
| |||
Litchfield |
| — | (3) |
| 2,200,000 |
|
| — |
| |||
Clyburn |
| — | (3) |
| 4,300,000 |
|
| — |
| |||
DeLuca |
| — | (3) |
| 2,700,000 |
|
| — |
| |||
Li |
| — | (3) |
| 3,000,000 |
|
| — |
| |||
| Target Grant Value(1)
| Increase in Target Grant Value | |||||||||||
Executive Officer |
2022 |
2023 | ||||||||||
Davis | $11,750,000 | $13,500,000 | +$1,750,000 | |||||||||
Litchfield | 2,750,000 | 4,250,000 | +1,500,000 | |||||||||
Chattopadhyay | — | (2) | 3,300,000 | — | ||||||||
DeLuca | 3,000,000 | (2) | 3,200,000 | +200,000 | ||||||||
Li | 3,900,000 | 5,600,000 | +1,700,000 | |||||||||
| (1) | Grant values shown above will be different from the values shown in the Summary Compensation and Grants of Plan-Based Awards tables based on the fair value on grant date in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718 (“FASB ASC Topic |
| (2) | Mr. |
|
PSU Program
At the beginning of each year, we review the design of our PSU program to ensure that our metrics are focused on the long-term measures that are most applicable to driving value for the Company and its shareholders over a three-year performance period. Payouts under the PSU program are formulaic and, as such, the C&MD Committee does not consider individual performance or use discretion when determining final awards.
Financial targets applicable to the PSUs are established based on our three-year financial plan, which considers a variety of factors, including management, Board, and external expectations and aspirations of our long-term performance. The financial targets for our currently outstanding PSUs are EPS and R-TSR. R-TSR performance versus our primary peer group is measured at the end of the three-year period and compares Merck’s average annual TSR to the median TSR of our pharmaceuticalthat peer group. Each percentage point of outperformance or underperformance versus the median modifies the earned award up or down by +/-5five percentage points. In the event of underperformance by more than 10 percentage points, there will not be a payout on the R-TSR portion of the award. In the event of outperformance, the payout
Merck & Co., Inc. 2022 Proxy Statement
|
on the R-TSR portion of the award cannot exceed 200%. If R-TSRour annualized TSR is negative, the payout on this portion of the award cannot exceed 100%, even if our R-TSR outperforms the median of the peer group.
Merck & Co., Inc. 2024 Proxy Statement
| 58 | ç ç ç ç | Compensation Discussion and Analysis The Elements of 2023 Compensation |
The 20192021 PSU program endedconcluded at the end of 20212023 and the payout is described on the following page. Beginning in 2020, we removed the OCF metric and increased the weighting of EPS to streamline our program design, focusing on a single earnings metric. Additionally, duebelow. Due to the complexities associated with disentangling our Organon business from a multi-year financial plan, we adjusted the design for the 2019, 2020, and 2021 programsprogram as further described below. Our Organon business was successfully spun off in June 2021. With having completedFollowing the completion of the spin-off, in 2021 as planned, we will revertreverted to a three-year cumulative EPS and R-TSR design inbeginning with the 2022 PSU program, with 50% tied to EPS and 50% tied to R-TSR.
Program Performance Period | Original Program Design | Program Design as a result of the Organon Spin-off | |||
|
|
| |||
|
|
| |||
2021-2023 | 50% 3-Year EPS 50% 3-Year R-TSR | 33% 1-Year (2021) EPS 67% 3-Year R-TSR | |||
| 2022-2024 2023-2025 | 50% 3-Year EPS | Not Applicable | |||
| (1) | Alternative design was established on grant date and part of original grant terms. |
Merck & Co., Inc. 2022 Proxy Statement
|
|
Payouts Under the 2019–20212021–2023 PSU Program Performance Period
For grants issued in 2019,2021, 70% of each NEO’s annual target LTI was converted to PSUs based on the closing price of Merck stock on the date of grant. The number of PSUs ultimately earned is based on our performance against the pre-established EPS target and OCF targets and R-TSR performance. As a result of the spin-off of Organon, the original number of PSUs granted were adjusted to preserve the same intrinsic value as was in place immediately prior to the adjustments.
For the 2019-20212021-2023 performance period, as a result of the Organon spin-off, two-year (2019-2020) cumulativeone-year (2021) EPS and OCF metrics were eachwas weighted at 25%33%, and three-year R-TSR versus our pharmaceuticalprimary peer group was weighted at 50%67%. If the Organon spin-off did not occur, three-year cumulative EPS and OFC metrics would have been used. The outcome of the combined performance resulted in an actual payout of 140%161% as illustrated in the tables below.
The 140%161% payout was based on our strong EPS and OCFR-TSR performance (both at 200%above 150%) during the performance period due to the momentumEmergency Use Authorization of LAGEVRIO in key growth areas of the business that delivered above-plan after-tax non-GAAP net income. While exceedingUnited States in 2021 and our operational metrics, we underperformedTSR outperforming the median TSR of our pharmaceuticalprimary peer group by 4 percentage points, which decreased the payout by 5% for each percentage point of underperformance, resulting in an R-TSR payout of 80%nearly 11%.
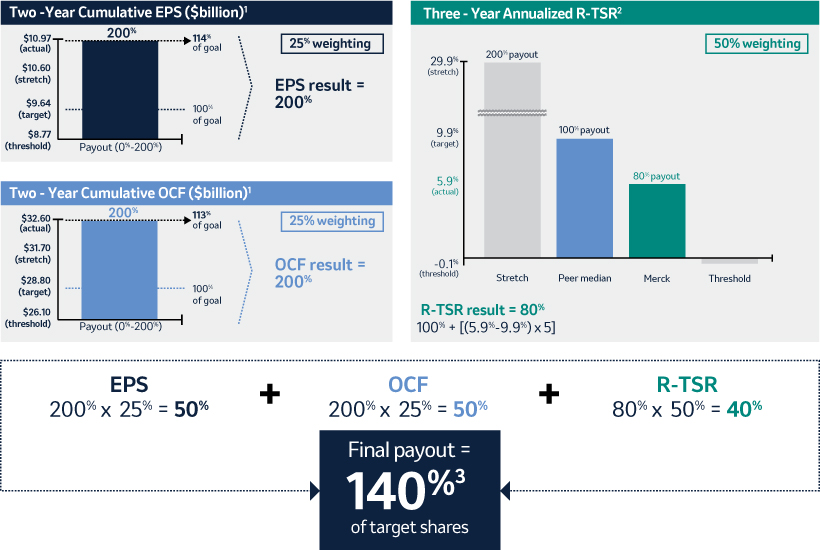
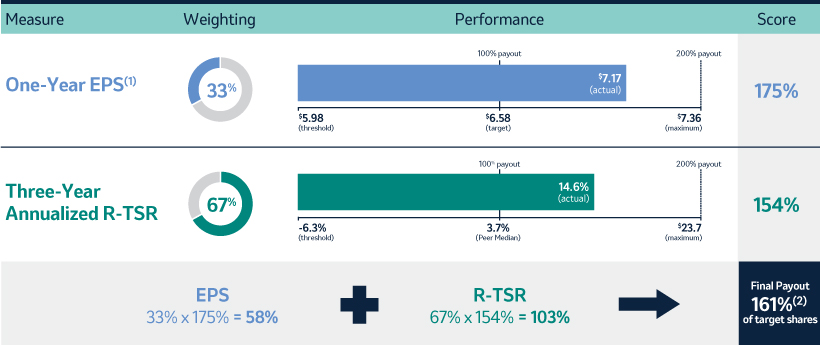
| (1) |
|
| (2) |
|
Rounded to the nearest whole percentage. |
Merck & Co., Inc. 2022 2024 Proxy Statement
Compensation Discussion and Analysis The Elements of | ÷ ÷ ÷ ÷ | 59 |
Named Executive Officer PSU Distribution
Based on the final payout of 140%161%, the NEOs received the following number of shares of Merck common stock, including dividends accrued during the performance period and paid in shares:
Named Executive Officer | Pre-Spin Target Award (# of shares) | Adjusted Post-Spin (# of shares) | Final Award(2) (# of shares) | Pre-Spin Target Award (# of shares) | Adjusted Post-Spin Target Award(1) (# of shares) | Final Award(2) (# of shares) | ||||||||||||||||||
Davis | 33,666 | 34,699 | 53,457 | |||||||||||||||||||||
Frazier | 126,247 | 130,120 | 200,456 | |||||||||||||||||||||
Davis | ||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||
Davis | 83,539 | 86,102 | 148,374 | |||||||||||||||||||||
Litchfield | 2,886 | 2,975 | 4,584 | |||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||
Litchfield | 19,977 | 20,590 | 35,481 | |||||||||||||||||||||
Clyburn | 15,150 | 15,615 | 24,056 | |||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||
Chattopadhyay | 25,425 | 26,205 | 45,157 | |||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||
DeLuca | 16,833 | 17,349 | 26,728 | 24,517 | 25,269 | 43,544 | ||||||||||||||||||
Li | 2,886 | 2,975 | 4,584 | |||||||||||||||||||||
Li | ||||||||||||||||||||||||
Li | ||||||||||||||||||||||||
Li | 27,241 | 28,077 | 48,384 | |||||||||||||||||||||
| (1) | As a result of the Organon spin-off, |
| (2) | Includes accrued dividends distributed in shares following final award determination. |
Additional information regarding the payouts under the 2019-20212021-2023 PSU performance period is provided in the Option Exercises and Stock Vested table on page 71.77.
To accurately reflect the operating performance of our business, the C&MD Committee approved a consistent framework of adjustments to our reported financial results for incentive program purposes. For further explanation of these adjustments and our GAAP versus Non-GAAP results, please refer to Appendices A and B on pages 97104 and 99,106, respectively.
In connection with Merck’s 2021 CEO transition, the C&MD Committee approved retention LTI grants for Mr. Clyburn ($3,000,000) and Mr. DeLuca ($2,000,000). Under their leadership, the Human Health and Animal Health businesses delivered strong results in 2020, despite the impact of the COVID-19 pandemic, in what continues to be a highly competitive and challenging marketplace. These two retention grants were intended to ensure Mr. Clyburn and Mr. DeLuca remained focused on leading their respective businesses during and following the CEO transition. The LTI grants were issued on May 4, 2021 in the form of 100% RSUs, vesting in their entirety at the end of a three-year period, subject to their continued employment. As a result of Mr. Clyburn’s resignation, effective February 1, 2022, he forfeited the full value of his retention LTI grant.
| Retention LTI Grant | ||||||||||||
Named Executive Officer | RSU Retention Grant Value | Grant Date | Vest Date | |||||||||
Clyburn | $ | 3,000,000 | May 4, 2021 | Forfeited | ||||||||
DeLuca | 2,000,000 | May 4, 2021 | May 4, 2024 | |||||||||
Similar to Merck’s other salaried, U.S.-based employees, the NEOs participate in a variety of retirement, health and welfare, and paid time-off benefits designed to enable us to attract and retain our workforce in a highly competitive market. Pension and savings plans help employees save and prepare financially for retirement. Health and welfare and paid time-off benefits help ensure that we have a healthy, productive, and focused workforce.
Additionally, senior management employees, including the NEOs, are provided a limited number of other benefits, which the C&MD Committee believes are reasonable, appropriate, and consistent with our executive compensation philosophy.
These benefits, which are described in more detail below, are reflected in the “All Other Compensation” column of the Summary Compensation table.
| • | Financial and tax planning. Executives receive a $10,000 cash allowance each December to encourage consultation with knowledgeable financial and tax planning experts who can help them understand the compensation and benefits programs in which they participate. |
Financial and tax planning. Executives receive a $10,000 cash allowance each December to encourage consultation with knowledgeable financial and tax planning experts who can help them understand the compensation and benefits programs in which they participate.
| • | Personal use of Company aircraft. Our global security organization regularly evaluates the travel risk for our CEO. As a result of these assessments and based on our security team’s recommendation, our Board of Directors has determined that our CEO must use Company-provided aircraft for all business and personal travel. Personal use of Company aircraft by other executives requires CEO approval and is only permitted under exceptional circumstances. Other than our CEO, there was no such reported usage for any other NEO in 2023. |
| • | Personal use of Company car and driver. Our CEO is provided with a car and driver to ensure his individual safety and security. Personal use of a car and driver is also provided to a select number of other executives, primarily for commuting purposes, allowing them to devote additional time to critical Company business. In 2023, personal use of a company car and driver was provided to Mr. Chattopadhyay and Dr. Li. |
| • | Residential security systems. Reimbursement for the installation, maintenance and remote access of residential security systems is provided to select executives, when deemed necessary by our internal global security team. Executives are responsible for paying monthly security monitoring fees, which are not reimbursable. Other than for our CEO, there was no reimbursement for any other NEO in 2023. |
Merck & Co., Inc. 2022 2024 Proxy Statement
| 60 | ç ç ç ç |
Compensation Discussion and Analysis The Elements of |
Personal use of Company aircraft. Our global security organization regularly evaluates the travel risk for our CEO. As a result of these assessments and based on our security team’s recommendation, our Board of Directors has determined that our CEO must use Company-provided aircraft for all business and personal travel. In addition, this requirement continued for Mr. Frazier following his transition to Executive Chairman, as a result of our security team’s recommendation. Personal use of Company aircraft by other executives requires CEO approval and is only permitted under exceptional circumstances. Other than our CEO and Executive Chairman, there was no reported usage for any other NEO.
Personal use of Company car and driver. Our CEO and Executive Chairman are provided with a car and driver to ensure their individual safety and security. Personal use of a car and driver is also provided to a select number of other executives, primarily for commutation purposes, allowing them to devote additional time to critical Company business.
Residential security systems. Reimbursement for the installation, maintenance and remote access of residential security systems is provided to select executives, when deemed necessary by our internal global security team. Executives are responsible for paying monthly security monitoring fees, which are not reimbursable.
As part of our annual compensation review, the C&MD Committee reviewed and approved target TDC opportunities for oureach executive officers,officer, including our NEOs.NEOs, for 2024. The Board of Directors (excluding Mr. Davis and Mr. Frazier)Davis) reviewed and approved Mr. Davis’ and Mr. Frazier’s target TDC.
Mr. Davis’Davis’s target TDC for 20222024.
Mr. Davis’s target TDC for 2024 increased by 7.7%11.4% based on a review of his performance and the competitive positioning of his compensation relative to the primary pharmaceutical and supplemental (Dow) peer groups. Consistent with our compensation strategy that supports a pay-for-performance culture, the Board intends to increasecontinue to adjust Mr. Davis’Davis’s target TDC, over timeand when appropriate, increase it to achieve a moreensure it remains competitive position relative to these peer groups assuming continued strongand aligned with the Board’s assessment of his business performance and leadership. As part of Mr. Davis’Davis’s target TDC increase, annual base salary increased by 3%, there was no change in target annual incentive percent, and target LTI was increased by $1,000,000 to $11,750,000.
Mr. Frazier’s 2022 target TDC was reduced by 43.4%, recognizing his reduced responsibilities as Executive Chairman for 2022 and consistent with market practice for CEOs transitioning to Executive Chairmen. No change was made to Mr. Frazier’shis annual base salary or target annual cash incentive percent. Mr. Frazier’sHis target LTI grant was reducedincreased by $5,750,000$2,000,000 to $5,000,000. In addition, while there is no definitive timing for Mr. Frazier’s retirement as Executive Chairman, the Board determined that his continued service as Executive Chairman would be for a transition period and, as such, intends to provide Mr. Frazier’s LTI grant in the form of RSU awards that vest one year from the date of the grant. Similar to Mr. Frazier’s 2019-2021 grants, the 2022 RSU awards will be subject to continued compliance with non-compete and non-solicit requirements.$15,500,000.
To better align their compensation with the overall market, Ms. Litchfield’s, Mr. DeLuca’s and Dr. Li’sThe target TDC for our other NEOs increased by 17.5%between 1.3% and 1.5%, 9.3% and 23.5%, respectively. This included market adjustmentsreflecting an increase of 4% to their annual base salaries and increases insalaries. There were no changes to their target annual cash incentive percentages or target LTI grants. Given Mr. Clyburn’s departure, he did not receive an increase to his annual base salary or an LTI grant.
The following table summarizes adjustments made to CEO Executive Chairman, and other NEO compensation for 2022.2024.
Named Executive Officer(1) | Target Total Direct Compensation Increase % | Annual Base Salary Increase% | Target Annual Incentive % of Base Salary | Target LTI Grant Value Increase $ | ||||||||||||
Davis | +7.7 | % | +3.0 | % | No change | +$1,000,000 | (2) | |||||||||
Frazier | -43.4 | No change | No change | -5,750,000 | ||||||||||||
Litchfield | +17.5 | +8.3 | (3) | No change | +550,000 | |||||||||||
DeLuca | +9.3 | +6.3 | (3) | No change | +300,000 | |||||||||||
Li | +23.5 | +13.2 | (3) | No change | +900,000 | |||||||||||
|
|
|
Merck & Co., Inc. 2022 Proxy Statement
Compensation Increase % |
| Target Annual Incentive % of | Target LTI Grant Value Increase $ | |||||||||||||
Davis | +11.4 | % | No Change | No change | +$2,000,000 | |||||||||||
Litchfield | +1.4 | +4.0 | % | No change | No change | |||||||||||
Chattopadhyay | +1.5 | +4.0 | No change | No change | ||||||||||||
DeLuca | +1.5 | +4.0 | No change | No change | ||||||||||||
Li | +1.3 | +4.0 | No change | No change | ||||||||||||
Stock Ownership Requirements
The C&MD Committee recognizes the critical role that executive stock ownership has in aligning the interests of management with those of shareholders. As such, we maintain a formal stock ownership policy under whichthat requires the CEO and other senior executives are required to acquire and hold a certain amount of Merck common stock in an amount representing a multiple of their base salary for so long as they remain in office. The amount of Merck common stock an executive is required to hold is based on a designated multiple of the executive’s base salary. Until the designated multiple of base salary is reached, executives are required to retain in stock a percentage of the after-tax net proceeds associated with stock option exercises and/or PSU and RSU settlements (100% for the CEO and Executive Chairman and 75% for the other NEOs). In calculating the attainment of our stock ownership requirements, we exclude (1) unexercised stock options and (2) unvested PSUs and RSUs. All NEOs have met their stock ownership requirements.
Merck & Co., Inc. 2024 Proxy Statement
Compensation Discussion and Analysis The Elements of 2023 Compensation | ÷ ÷ ÷ ÷ | 61 |
The following table sets forth the stock ownership requirements and current stock ownership status as a percentagemultiple of the requirementbase salary for the CEO Executive Chairman, and other NEOs as of February 28, 2022.29, 2024.
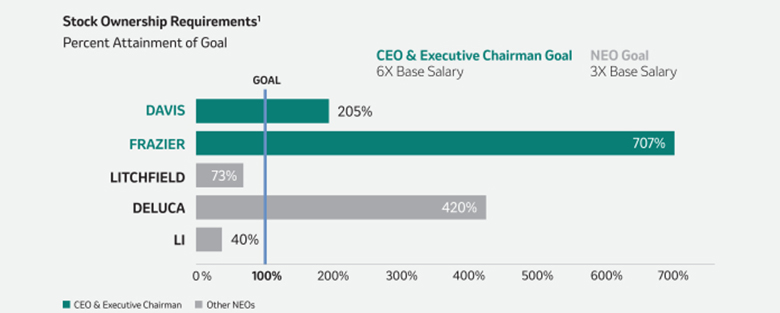

|
Return of Incentive Compensation (“Clawback Policy”)
Under our incentive compensation recoupmentThe C&MD Committee adopted a clawback policy, effective December 1, 2023, in compliance with Rule 10D-1 of the Board will seek reimbursement forSecurities Exchange Act of 1934 and Section 303A.14 of the annual cash incentive and/or LTI awards paidNew York Stock Exchange Listed Company Manual (the “Dodd-Frank Clawback Policy”).
In addition to the executive,Dodd-Frank Clawback Policy, executives are also subject to the recoupment of incentive-based cash compensation, equity-based compensation and any proceeds or earnings received in respect to such compensation, where the BoardC&MD Committee determines that (a) the executive engaged in misconduct or failed to reasonably supervise an employee who engaged in misconduct, that resulted in a (i) material violation of a written Company policy relating to (i) the research, development, manufacturing, sales or marketing of the Company’s products or (ii) conduct detrimental to the Company, including the Company’s overall goodwill or reputation ofto the Company, (the latter of which was added and approved by the C&MD Committee in November 2021), or (b) a significant restatement ofnegative impact on the Company’s financial operating results hasor reputation occurred. In the event of a financial restatement, the portion of the annual cash incentive and/or PSUs paid to the executive, in excess of the amount that would have been paid if the financial results were reported accurately, will be recouped.
Hedging and Pledging
As part of our insider trading policy, Merck prohibits Directors and management level employees, including officers, from engaging in short sales, publicly traded options, hedging transactions, and pledging of CompanyMerck common stock.
Tax Deductibility of Compensation
In light of the repeal of the performance-based compensation exemption under Section 162(m) of the Internal Revenue Code, the C&MD Committee may authorize compensation that is not deductible if it is determined to be appropriate and in the best interests of the Company and our shareholders.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 62 | ç ç ç ç |
Compensation Discussion and Analysis Compensation Risk Assessment |
Our executive compensation program and policies are driven by our business environment and designed to enable us to achieve our mission and adhere to our values. The C&MD Committee and senior management continually evaluate the relationship between risk and reward as it relates to our executive compensation program and have adopted policies and practices that mitigate undue risk while preserving the incentive/variable nature of the compensation. These policies and practices are described in more detail in the Compensation Policies and Practices chart on page 46.48.
In 2020,2022, Merck engaged Pay Governance, a compensation consultant to management, to perform a formal assessment of our executive compensation program, policies, and practices based on generally accepted compensation practices. The results of the assessment were reviewed by FW Cook, the C&MD Committee’s independent compensation consultant, and then discussed with the C&MD Committee in November 2020.2022. The assessment reaffirmed our belief that our compensation programs and policies are structured and operated in a manner that does not create risks that are reasonably likely to have a material adverse effect on our business. In addition to ongoing monitoring of our programs and policies, we are committed to performing formal assessments on a periodic basis. The next formal assessment is scheduled for review and discussion with the C&MD Committee in November 2022.2024.
Compensation and Management Development Committee Report
The C&MD Committee, comprised of independent Directors, reviewed and discussed the above CD&A with management. Based on the review and discussions, the C&MD Committee recommended to our Board of Directors that the CD&A be included in these proxy materials.
Compensation and Management Development Committee
Patricia F. Russo (Chair)
Mary Ellen Coe
Thomas H. Glocer
Risa J. Lavizzo-Mourey, M.D.
Inge G. Thulin
Peter C. Wendell
Merck & Co., Inc. 2022 2024 Proxy Statement
| ÷ ÷ ÷ ÷ | 63 |
The following table summarizes the total compensation that was paid or accrued for the Named Executive Officers for the fiscal years ended December 31, 2021, 2020,2023, 2022, and 2019.2021. The Named Executive Officers are the Company’s Chief Executive Officer, Executive Chairman, Chief Financial Officer and the three next most highly compensated executive officers as of December 31, 2021.2023. All amounts in the following table are rounded to the nearest dollar.
Name and Principal Position | Year | Salary ($)(1) | Bonus ($) | Stock Awards ($)(2) | Option Awards ($)(3) | Non-Equity Incentive Plan Compensation ($)(4) | Change in Pension Value and Nonqualified Deferred Compensation Earnings ($)(5) | All Other Compensation ($)(6) | Total ($) | |||||||||||||||||||||||||||
Robert M. Davis Chief Executive Officer, President and Former Chief Financial Officer |
| 2021 |
| $ | 1,319,959 |
|
| 0 |
|
| $6,324,576 |
| $ | 2,760,003 |
|
| $2,834,606 |
|
| $235,640 | (7) |
| $247,337 |
| $ | 13,722,121 |
| |||||||||
| 2020 |
|
| 1,112,795 |
|
| 0 |
|
| 2,737,406 |
|
| 1,199,772 |
|
| 1,018,194 |
|
| 611,948 |
|
| 159,395 |
|
| 6,839,510 |
| ||||||||||
| 2019 |
|
| 1,075,557 |
|
| 0 |
|
| 3,046,785 |
|
| 1,200,201 |
|
| 2,090,702 |
|
| 193,079 |
|
| 120,864 |
|
| 7,727,188 |
| ||||||||||
Kenneth C. Frazier Executive Chairman and Former Chief Executive Officer |
| 2021 |
|
| 1,478,681 |
|
| 0 |
|
| 7,390,092 |
|
| 3,225,004 |
|
| 2,804,094 |
|
| 0 | (8) |
| 299,049 |
|
| 15,196,920 |
| |||||||||
| 2020 |
|
| 1,702,006 |
|
| 0 |
|
| 10,778,499 |
|
| 4,724,098 |
|
| 2,218,500 |
|
| 2,288,641 |
|
| 376,685 |
|
| 22,088,429 |
| ||||||||||
| 2019 |
|
| 1,659,482 |
|
| 0 |
|
| 11,425,398 |
|
| 4,500,763 |
|
| 4,609,200 |
|
| 5,078,147 | (9) |
| 375,485 |
|
| 27,648,475 |
| ||||||||||
Caroline Litchfield Executive Vice President and Chief Financial Officer |
| 2021 |
|
| 805,060 |
|
| 0 |
|
| 1,512,420 |
|
| 660,001 |
|
| 1,184,203 |
|
| 0 | (13) |
| 59,770 |
|
| 4,221,454 |
| |||||||||
| 2020 | (10) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
| 2019 | (10) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
Frank Clyburn Former Executive Vice President and President, Human Health(11) |
| 2021 |
|
| 960,343 |
|
| 0 |
|
| 5,956,031 | (12) |
| 1,290,003 |
|
| 1,443,506 |
|
| 187,604 |
|
| 83,584 |
|
| 9,921,071 |
| |||||||||
| 2020 | (10) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
| 2019 | (10) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
Richard R. DeLuca, Jr. Executive Vice President and President, Merck Animal Health |
| 2021 |
|
| 790,247 |
|
| 0 |
|
| 3,856,115 | (12) |
| 809,999 |
|
| 1,184,000 |
|
| 128,732 |
|
| 71,907 |
|
| 6,841,000 |
| |||||||||
| 2020 | (10) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
| 2019 | (10) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
Dean Li, M.D., Ph.D. Executive Vice President and President, Merck Research Laboratories |
| 2021 |
|
| 937,104 |
|
| 0 |
|
| 2,062,363 |
|
| 900,003 |
|
| 1,400,799 |
|
| 114,593 |
|
| 66,057 |
|
| 5,480,919 |
| |||||||||
| 2020 | (10) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
| 2019
| (10)
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
Name and Principal Position | Year | Salary | Bonus | Stock | Option | Non-Equity | Change in | All Other | Total | |||||||||||||||||||||||||||
Robert M. Davis Chairman, Chief Executive Officer and President |
| 2023 |
| $ | 1,603,091 |
|
| $0 |
|
| $9,997,585 |
| $ | 4,050,000 |
|
| $3,585,300 |
|
| $651,163 | (7) |
| $386,148 |
| $ | 20,273,287 |
| |||||||||
| 2022 |
|
| 1,538,613 |
|
| 0 |
|
| 8,868,587 |
|
| 3,524,995 |
|
| 4,125,150 |
|
| 180,259 | (8) |
| 412,490 |
|
| 18,650,093 |
| ||||||||||
| 2021 |
|
| 1,319,959 |
|
| 0 |
|
| 6,324,576 |
|
| 2,760,003 |
|
| 2,834,606 |
|
| 235,640 |
|
| 247,337 |
|
| 13,722,121 |
| ||||||||||
Caroline Litchfield Executive Vice President and Chief Financial Officer |
| 2023 |
|
| 1,093,063 |
|
| 0 |
|
| 3,147,375 |
|
| 1,275,003 |
|
| 1,665,000 |
|
| 792,534 | (9) |
| 327,410 |
|
| 8,300,385 |
| |||||||||
| 2022 |
|
| 959,959 |
|
| 0 |
|
| 2,075,595 |
|
| 824,999 |
|
| 1,735,500 |
|
| 0 | (10) |
| 326,605 |
|
| 5,922,657 |
| ||||||||||
| 2021 |
|
| 805,060 |
|
| 0 |
|
| 1,512,420 |
|
| 660,001 |
|
| 1,184,203 |
|
| 0 | (11) |
| 434,946 |
|
| 4,596,630 |
| ||||||||||
Sanat Chattopadhyay Executive Vice President and President, Merck Manufacturing Division |
| 2023 |
|
| 934,923 |
|
| 0 |
|
| 2,443,907 |
|
| 989,997 |
|
| 1,393,873 |
|
| 317,466 |
|
| 160,194 |
|
| 6,240,360 |
| |||||||||
| 2022 | (14) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
| 2021 | (14) |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||||||
Richard R. DeLuca, Jr. Executive Vice President and President, Merck Animal Health |
| 2023 |
|
| 910,027 |
|
| 0 |
|
| 2,369,846 |
|
| 959,999 |
|
| 1,369,000 |
|
| 470,087 | (12) |
| 118,931 |
|
| 6,197,890 |
| |||||||||
| 2022 | (14) |
| 840,522 |
|
| 0 |
|
| 2,264,301 |
|
| 899,993 |
|
| 1,513,000 |
|
| 0 | (13) |
| 101,011 |
|
| 5,618,828 |
| ||||||||||
| 2021 |
|
| 790,247 |
|
| 0 |
|
| 3,856,115 | (15) |
| 809,999 |
|
| 1,184,000 |
|
| 128,732 |
|
| 71,907 |
|
| 6,841,000 |
| ||||||||||
Dean Li, M.D., Ph.D. Executive Vice President and President, Merck Research Laboratories |
| 2023 |
|
| 1,368,819 |
|
| 0 |
|
| 4,147,202 |
|
| 1,679,999 |
|
| 2,072,000 |
|
| 339,249 |
|
| 172,146 |
|
| 9,779,414 |
| |||||||||
| 2022 |
|
| 1,095,055 |
|
| 0 |
|
| 2,943,574 |
|
| 1,169,998 |
|
| 2,225,000 |
|
| 210,204 |
|
| 122,233 |
|
| 7,766,063 |
| ||||||||||
| 2021 |
|
| 937,104 |
|
| 0 |
|
| 2,062,363 |
|
| 900,003 |
|
| 1,400,799 |
|
| 114,593 |
|
| 66,057 |
|
| 5,480,919 |
| ||||||||||
| (1) | Amounts shown reflect actual base salary earnings and are not reduced to reflect the Named Executive Officers’ elections, if any, to defer receipt of salary into the Merck Deferral Program, an unfunded nonqualified savings plan. |
For more information about deferred amounts, see the Nonqualified Deferred Compensation table and related footnotes on page |
| (2) | The amounts shown in this column represent the full grant date fair value of RSUs and PSUs granted to each of the Named Executive Officers during |
The maximum value of the PSU awards granted to the Named Executive Officers during |
Named Executive Officer | Maximum Value of PSU Awards ($) | |||
Davis | $ | 12,649,152 |
| |
Frazier |
| 14,780,183 |
| |
Litchfield |
| 3,024,840 |
| |
Clyburn |
| 5,912,043 |
| |
DeLuca, Jr. |
| 3,712,269 |
| |
Li |
| 4,124,727 |
| |
Named Executive Officer | Maximum Value of PSU Awards ($) | |||
Davis | $19,995,171 | |||
Litchfield | 6,294,751 | |||
Chattopadhyay | 4,887,813 | |||
DeLuca | 4,739,691 | |||
Li | 8,294,403 | |||
For more information on the awards granted during |
Merck & Co., Inc. 2022 Proxy Statement
|
|
| (3) | The amounts shown in this column represent the full grant date fair value of stock options granted to each of the Named Executive Officers during |
Merck & Co., Inc. 2024 Proxy Statement
| 64 | ç ç ç ç | Summary Compensation Table |
For more information on stock options granted during |
| (4) | Represents amounts paid under the |
Amounts shown are not reduced to reflect the Named Executive Officers’ elections, if any, to defer receipt of awards into the Merck Deferral Program. For more information, see the Nonqualified Deferred Compensation table and related notes and narrative on page |
| (5) | Amounts shown are solely an estimate of the aggregate change in actuarial present value of the Named Executive Officers’ accrued benefits under the Company’s pension plans from December 31, |
The Merck Deferral Program, an unfunded nonqualified savings plan, does not provide for above market or preferential earnings. For more information, see the Nonqualified Deferred Compensation table and related notes and narrative on page |
| (6) | See the All Other Compensation table on page |
| (7) |
|
| (8) | For 2022, the change in value compared to the previous year is |
|
| (10) | For 2022, the U.S. change in pension value is positive; however, the U.K. change in pension value is negative primarily due to |
|
|
| (11) |
|
|
|
| (12) | For 2023, the change in value for Mr. DeLuca is primarily due to a decrease in discount rates as of December 31, 2023. |
| (13) | For 2022, the change in value for Mr. DeLuca is negative primarily due to an increase in discount rates. In accordance with SEC rules, a $0 value is reported rather than a negative amount. |
| (14) | Mr. Chattopadhyay was not a Named Executive Officer in 2021 and 2022. Mr. DeLuca was not a Named Executive Officer in 2022. While Mr. DeLuca was not a Named Executive Officer in 2022, he was a Named Executive Officer in 2021. As such, pursuant to SEC rules, we have included Mr. DeLuca’s 2022 compensation information in this Summary Compensation table. |
| (15) | Includes value of $2,000,000 RSU retention grant that will vest on May 4, 2024, subject to his continued employment. |
Merck & Co., Inc. 2022 2024 Proxy Statement
Summary Compensation Table | ÷ ÷ ÷ ÷ | 65 |
All Other Compensation
Name | Year | Financial/Tax Counseling & Tax Preparation Services ($)(1) | Company Aircraft ($)(2) | Company Car and Driver ($)(3) | Installation, Maintenance and Remote Access ($)(4) | Relocation Expense ($) | Savings Plan Company Match and Credits ($)(5) | Total ($) | ||||||||||||||||||||||||
Davis | 2021 | $10,000 | $94,552 | $11,353 | $26,258 | $0 | $105,174 | $247,337 | ||||||||||||||||||||||||
| 2020 | 10,000 | 0 | 3,620 | 0 | 0 | 145,775 | 159,395 | ||||||||||||||||||||||||
|
| 2019 | 10,000 | 0 | 0 | 0 | 0 | 110,864 | 120,864 | ||||||||||||||||||||||||
Frazier | 2021 | 10,000 | 93,769 | 25,262 | 3,966 | 0 | 166,052 | 299,049 | ||||||||||||||||||||||||
| 2020 | 10,000 | 21,983 | 50,572 | 7,637 | 0 | 286,493 | 376,685 | ||||||||||||||||||||||||
|
| 2019 | 10,000 | 90,661 | 50,121 | 12,378 | 0 | 212,325 | 375,485 | ||||||||||||||||||||||||
Litchfield | 2021 | 10,000 | 0 | 0 | 0 | 0 | 49,770 | 59,770 | ||||||||||||||||||||||||
| 2020 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
|
| 2019 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
Clyburn | 2021 | 10,000 | 0 | 1,358 | 0 | 0 | 72,226 | 83,584 | ||||||||||||||||||||||||
| 2020 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
|
| 2019 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
DeLuca, Jr. | 2021 | 10,000 | 0 | 0 | 0 | 0 | 61,907 | 71,907 | ||||||||||||||||||||||||
| 2020 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
|
| 2019 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
Li | 2021 | 10,000 | 0 | 0 | 0 | 0 | 56,057 | 66,057 | ||||||||||||||||||||||||
| 2020 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
|
| 2019 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
Name | Year | Financial/Tax Counseling & Tax Preparation Services ($)(1) | Company Aircraft ($)(2) | Company Car and Driver ($)(3) | Installation, Maintenance and Remote Access of Home Security ($)(4) | Relocation Expense and Tax Equalization ($) | Savings Plan Company Match and Credits ($)(5) | Total ($) | ||||||||||||||||||||||||
Davis | 2023 | $10,000 | $91,519 | $15,224 | $11,824 | $0 | $257,580 | $386,148 | ||||||||||||||||||||||||
| 2022 | 10,000 | 191,073 | 8,113 | 6,689 | 0 | 196,615 | 412,490 | ||||||||||||||||||||||||
|
| 2021 | 10,000 | 94,552 | 11,353 | 26,258 | 0 | 105,174 | 247,337 | ||||||||||||||||||||||||
Litchfield | 2023 | 10,000 | 0 | 0 | 0 | 190,245 | (7) | 127,165 | 327,410 | |||||||||||||||||||||||
| 2022 | 10,000 | 0 | 0 | 0 | 220,220 | (7) | 96,385 | 326,605 | |||||||||||||||||||||||
|
| 2021 | 10,000 | 0 | 0 | 0 | 375,176 | (7) | 49,770 | 434,946 | |||||||||||||||||||||||
Chattopadhyay | 2023 | 10,000 | 0 | 36,044 | 0 | 0 | 114,150 | 160,194 | ||||||||||||||||||||||||
| 2022 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
|
| 2021 | (6) | — | — | — | — | — | — | — | |||||||||||||||||||||||
DeLuca | 2023 | 10,000 | 0 | 0 | 0 | 0 | 108,931 | 118,931 | ||||||||||||||||||||||||
| 2022 | (6) | 10,000 | 0 | 0 | 0 | 0 | 91,011 | 101,011 | |||||||||||||||||||||||
|
| 2021 | 10,000 | 0 | 0 | 0 | 0 | 61,907 | 71,907 | ||||||||||||||||||||||||
Li | 2023 | 10,000 | 0 | 579 | 0 | 0 | 161,567 | 172,146 | ||||||||||||||||||||||||
| 2022 | 10,000 | 0 | 0 | 0 | 0 | 112,233 | 122,233 | ||||||||||||||||||||||||
|
| 2021 | 10,000 | 0 | 0 | 0 | 0 | 56,057 | 66,057 | ||||||||||||||||||||||||
| (1) | The Named Executive Officers receive a cash allowance each December for financial and tax planning benefits. |
| (2) | The value of any personal use of Company aircraft by the Named Executive Officers is based on the aggregate incremental per-hour cost based on the flight time flown from origination to destination and a return to point of origination without passengers, when applicable. This benefit is taxable to the Named Executive Officers. As further described in the Other Employee Benefits section on page |
| (3) | The value of any personal use of Company car and driver by the Named Executive Officers is based on the recipient’s cost if equivalent assets were used independent of the Company. This benefit is taxable to the Named Executive Officers. |
The incremental cost calculation for personal use of Company car and driver by the Named Executive Officers includes driver overtime, meals, travel pay, maintenance and fuel costs. Personal use of a car and driver is also provided to a select number of other executives, primarily for commuting purposes, as further described in the Other Employee Benefits section on page 59.
|
| (4) | Installation, maintenance, and remote access of home security are valued at actual costs billed by outside vendors. |
| (5) | The Named Executive Officers received Company matching contributions equal to 75% of the first 6% of eligible compensation contributed (up to the IRS limit for qualified savings plans) to the Merck U.S. Savings Plan and a 4.5% credit of eligible compensation in excess of the IRS limit to the Named Executive Officers’ accounts under the Merck Deferral Program. |
| (6) | Mr. Chattopadhyay was not a Named Executive Officer in 2021 and 2022. Mr. DeLuca was not a Named Executive Officer in 2022. While Mr. DeLuca was not a Named Executive Officer in 2022, he was a Named Executive Officer in 2021. As such, pursuant to SEC rules, we have included Mr. DeLuca’s 2022 compensation information in this All Other Compensation table. |
| (7) | Prior to her appointment as our Chief Financial Officer, Ms. Litchfield |
Merck & Co., Inc. 2022 2024 Proxy Statement
| 66 | ç ç ç ç |
|
Introduction
The following is a disclosure of (1) total annual compensation for our CEO, (2) the median total annual compensation for our employees globally, excluding our CEO and (3) the ratio of those two numbers.
Median Total Annual Compensation
We used base salary as of October 31, 2021,2023 to identify the employee with the median total annual compensation (not including(excluding our CEO). For this purpose, we annualized base salary for all full and part-time employees (other than(excluding our CEO) hired after January 1, 20212023 and employed as of October 31, 2021.2023. We converted foreign currency to U.S. dollars using a twelve-month average exchange rate between November 1, 20202022 through October 31, 2021.2023.
Exemptions
Total Employees Before and After De Minimis ExemptionEmployee Population
Merck’s employee population as of October 31, 20212023 included 26,810 (37%29,358 (39%) employees in the United States and 45,197 (63%46,859 (61%) employees outside the United States. After excluding 3,6003,810 employees in 1915 countries, as detailed in the table below and up to the 5% limit allowable under the SEC disclosure rules, we identified our median employee from a group of approximately 68,40772,407 employees globally.
Excluded Under De Minimis Exemption
Country | Number of Employees | Country | Number of Employees | |||||||||||
Algeria | 35 | Jordan | 23 | |||||||||||
Bosnia and Herzegovina | 8 | Latvia | 18 | |||||||||||
Colombia | 1,040 | Malaysia | 371 | |||||||||||
Dominican Republic | 7 | Nigeria | 1 | |||||||||||
Egypt | 185 | North Macedonia | 3 | |||||||||||
Honduras | 5 | Philippines | 206 | |||||||||||
India | 1,489 | Vietnam | 253 | |||||||||||
Indonesia | 166 | |||||||||||||
TOTAL | 3,810 | |||||||||||||
Country | Number of Employees | Country | Number of Employees | |||||||||||
Algeria | 60 | Jordan | 35 | |||||||||||
Belarus | 2 | Malaysia | 464 | |||||||||||
Bosnia and Herzegovina | 6 | Nigeria | 1 | |||||||||||
Bulgaria | 59 | Oman | 6 | |||||||||||
Dominican Republic | 11 | Peru | 147 | |||||||||||
Ecuador | 55 | Philippines | 230 | |||||||||||
Egypt | 196 | Turkey | 526 | |||||||||||
Honduras | 6 | Uruguay | 90 | |||||||||||
India | 1,224 | Vietnam | 282 | |||||||||||
Indonesia | 200 | |||||||||||||
TOTAL | 3,600 | |||||||||||||
The Ratio
The total annual compensation of our median employee as calculated under the Summary Compensation table requirements for calculating total annual compensation was $102,803$110,827 comprised of base salary, annual cash incentive, savings plan company match, and change in pension value. The total annual compensation for our CEO was $13,722,121.$20,273,287. A reasonable estimation of the ratio of our CEO’s compensation to our median employee’s compensation is 133183 to 1.
Under the SEC rules, companies may identify the median total annual compensation using a wide variety of methods, including reasonable assumptions and estimations. It is, therefore, difficult to compare Merck’s ratio to the ratios of other companies.
Merck & Co., Inc. 2022 2024 Proxy Statement
÷ ÷ ÷ ÷ | 67 |
Company Scorecard Metrics | PSU Program Metrics | |
| Revenue | Relative Total Shareholder Return | |
Pre-Tax Income | Non-GAAP Earnings Per Share (EPS) | |
| Pipeline | ||
| Sustainability | ||

| 68 | ç ç ç ç | Pay Versus Performance Pay Versus Performance Table |
Summary Compensation Table Total for CEO (Davis) | Summary Compensation Table Total for CEO (Frazier) | Compensation Actually Paid to CEO (Davis) (5) | Compensation Actually Paid to CEO (Frazier) (5) | Average Summary Compensation Table Total for Non-CEO NEOs | Average Compensation Actually Paid to Non-CEO NEOs (5) | Value of Initial Fixed $100 Investment Based on | ||||||||||||||||||||||||||||||||||
Fiscal Year | Total Shareholder Return (6) | Peer Group Total Shareholder Return (7) | GAAP Net Income ($ 000’s) | Non-GAAP EPS (8) | ||||||||||||||||||||||||||||||||||||
2023 (1) | $20,273,287 | — | $26,701,326 | — | $7,629,512 | $9,360,664 | $142.53 | $142.00 | $364,655 | $1.51 | ||||||||||||||||||||||||||||||
2022 (2) | 18,650,093 | — | 52,474,235 | — | 8,406,647 | 22,341,814 | 141.12 | 138.11 | 14,519,000 | 7.48 | ||||||||||||||||||||||||||||||
2021 (3) | 13,722,121 | 15,196,920 | 14,390,893 | 13,328,924 | 6,709,905 | 7,004,950 | 94.47 | 130.44 | 12,345,000 | 5.37 | ||||||||||||||||||||||||||||||
2020 (4) | — | 22,088,429 | — | 10,578,487 | 6,041,722 | 3,582,756 | 92.79 | 106.53 | 4,519,000 | 2.97 | ||||||||||||||||||||||||||||||
| (1) | Non-CEO NEOs for 2023 are Ms. Litchfield, Mr. Chattopadhyay, Mr. DeLuca, and Dr. Li. |
| (2) | Non-CEO NEOs for 2022 are Mr. Frazier, Ms. Litchfield, Mr. Guindo, Dr. Li, and Ms. Zachary. Since Mr. Davis was CEO for all of 2022, only his compensation is shown in the CEO SCT and CAP columns. |
| (3) | Non-CEO NEOs for 2021 are Ms. Litchfield, Mr. Clyburn, Mr. DeLuca, and Dr. Li. As a result of Mr. Frazier’s retirement as CEO, effective June 30, 2021, and Mr. Davis’s promotion to CEO, effective July 1, 2021, the Pay versus Performance table includes the compensation for both executives as CEO in 2021. The SCT and CAP amounts reported for Mr. Davis and Mr. Frazier reflect compensation for all of 2021, not just the respective periods in which they were CEO. |
| (4) | Non-CEO NEOs for 2020 are Mr. Davis, Mr. Chattopadhyay, Dr. Perlmutter, and Ms. Zachary. Since Mr. Frazier was CEO for all of 2020, only his compensation is shown in the CEO SCT and CAP columns. |
| (5) | See following table for additional details on the calculation of the CAP value. |
| (6) | TSR assumes an initial $100 investment in Merck stock beginning on December 31, 2019. TSR is cumulative, with the value determined at the end of each applicable fiscal year, calculated in accordance with Item 201(e) of Regulation S-K. |
| (7) | Peer Group TSR assumes an initial $100 investment in Merck’s primary peer group (market cap-weighted). As described on page 49, Merck’s primary peer group consists of the following companies: AbbVie, Amgen, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Gilead Sciences, GlaxoSmithKline, Johnson & Johnson, Novartis, Pfizer, Roche Holding AG, and Sanofi. |
| (8) | The SEC requires the disclosure of a company selected measure, representing the most important financial metric used for determining CAP for the current fiscal year. As described in more detail on page 71, the selected measure for 2023 is non-GAAP EPS. Refer to Appendix A on page 104 for a reconciliation between GAAP andnon-GAAP financial measures. Appendix B on page 106 provides an explanation of adjustments tonon-GAAP results for incentive plans purposes. |
Pay Versus Performance Pay Versus Performance Table | ÷ ÷ ÷ ÷ | 69 |
Summary Compensation Table Total Compensation | Value of Pension Benefits Deducted from SCT (1) | Value of Equity Deducted from SCT | Value of Pension Benefits Per CAP Definition (2) | Fair Value of Equity Compensation Granted in Current Year (3) | Year-Over-Year Change in Fair Value of Unvested Equity (4) | Change in Fair Value of Equity that Vested During the Year (5) | Value of Dividends Accrued or Paid on Stock Awards (6) | Compensation Actually Paid | ||||||||||||||||||||||||||||
CEO (Davis) | ||||||||||||||||||||||||||||||||||||
| 2023 | $20,273,287 | $651,163 | $14,047,586 | $271,130 | $18,028,095 | $(97,780) | $1,522,952 | $1,402,391 | $26,701,326 | |||||||||||||||||||||||||||
2022 | 18,650,093 | 180,259 | 12,393,581 | 280,085 | 26,824,642 | 14,171,478 | 3,931,065 | 1,190,712 | 52,474,235 | |||||||||||||||||||||||||||
2021 | 13,722,121 | 235,640 | 9,084,579 | 265,823 | 10,649,030 | (669,339 | ) | (661,118 | ) | 404,596 | 14,390,893 | |||||||||||||||||||||||||
CEO (Frazier) | ||||||||||||||||||||||||||||||||||||
2021 | 15,196,920 | — | 10,615,096 | 418,183 | 12,443,073 | (2,633,787 | ) | (2,435,189 | ) | 954,819 | 13,328,924 | |||||||||||||||||||||||||
2020 | 22,088,429 | 2,288,641 | 15,502,597 | 379,037 | 17,150,711 | (5,340,113 | ) | (7,290,425 | ) | 1,382,086 | 10,578,487 | |||||||||||||||||||||||||
Average Non-CEO NEOs | ||||||||||||||||||||||||||||||||||||
2023 | 7,629,512 | 479,834 | 4,253,332 | 152,186 | 5,458,528 | (43,471 | ) | 475,952 | 421,122 | 9,360,664 | ||||||||||||||||||||||||||
2022 | 8,406,647 | 42,041 | 5,514,804 | 122,447 | 9,133,936 | 6,336,440 | 3,346,846 | 552,343 | 22,341,814 | |||||||||||||||||||||||||||
2021 | 6,709,905 | 107,732 | 4,261,734 | 122,147 | 4,829,729 | (247,588 | ) | (208,749 | ) | 168,972 | 7,004,950 | |||||||||||||||||||||||||
2020 | 6,041,722 | 498,315 | 3,518,845 | 157,683 | 3,892,938 | (1,222,908 | ) | (1,583,911 | ) | 314,391 | 3,582,756 | |||||||||||||||||||||||||
| (1) | Represents the aggregate change in actuarial present value of the NEOs’ accrued benefits under the Company’s pension plans. The change in pension value for Mr. Frazier is negative in 2021, primarily due to age and an increase in discount rates. In accordance with SEC rules, a $0 value is reported rather than a negative amount. |
| (2) | These amounts represent the present value of expected pension benefit accruals earned in the current year and reflects assumptions used for financial statement reporting purposes. They do not reflect the change in the present value of the accumulated pension benefit due to changes in assumptions such as discount rate from year to year. |
| (3) | These amounts represent the fair value as of the indicated fiscal year-end of the outstanding and unvested stock and option awards granted during such fiscal year, calculated in accordance with the methodology used for financial reporting purposes. The fair value differs from the value in the SCT because for purposes of CAP the fair value for equity granted in the current year is determined as of the last day of the applicable year. Fair values in the SCT are determined as of the grant date. |
| (4) | These amounts represent the change in fair value during the indicated fiscal year of each stock and option award that was granted in a prior fiscal year and that remained outstanding and unvested as of the last day of the indicated fiscal year, calculated in accordance with the methodology used for financial reporting purposes and, for awards subject to performance-based vesting conditions, based on an estimate of the probable outcome of such performance-based vesting conditions as of the last day of the fiscal year. |
| (5) | These amounts represent the change in fair value, measured from the prior fiscal year-end to the vesting date, of each stock and option award that was granted in a prior fiscal year and which vested during the indicated fiscal year, calculated in accordance with the methodology used for financial reporting purposes. |
| (6) | These amounts represent the dollar value of any dividends or other earnings accrued or paid on stock or option awards in the applicable fiscal year or for awards that vested in the fiscal year, prior to the vesting date, that are not otherwise reflected in the fair value of such awards or included in any other component of total compensation for the applicable fiscal year. |
| 70 | ç ç ç ç | Pay Versus Performance Pay Versus Performance Table |
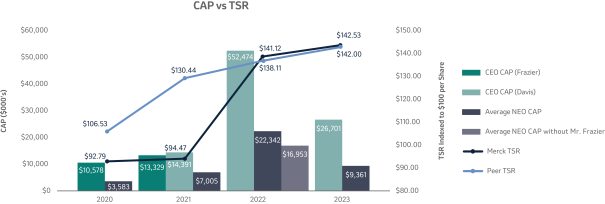
Reporting Year | Beginning | End | Number of Years | |||||||||||||||
| 2023 | 12/31/2019 | 12/31/2023 | 4 years | |||||||||||||||
| 2022 | 12/31/2019 | 12/31/2022 | 3 years | |||||||||||||||
| 2021 | 12/31/2019 | 12/31/2021 | 2 years | |||||||||||||||
| 2020 | 12/31/2019 | 12/31/2020 | 1 year | |||||||||||||||
Pay Versus Performance Pay Versus Performance Table | ÷ ÷ ÷ ÷ | 71 |
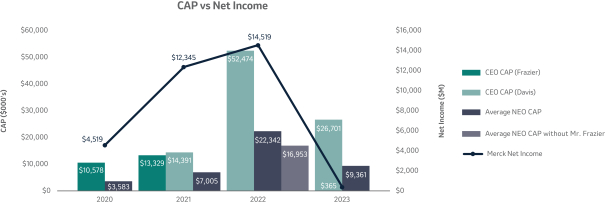
| 72 | ç ç ç ç | Pay Versus Performance Pay Versus Performance Table |
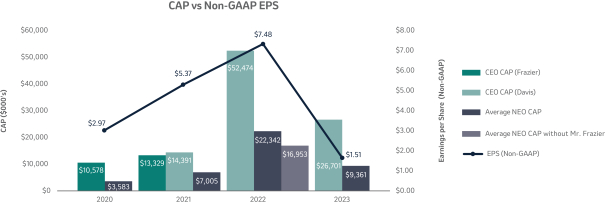
| ÷ ÷ ÷ ÷ | 73 |
The following table provides information concerning each award made in 20212023 to the Named Executive Officers under any incentive plan.
Grants of Plan-Based Awards for Fiscal Year Ended December 31, 20212023
| Estimated Future Payouts Under Non-Equity Incentive Plan Awards | Estimated Future Payouts Under Equity Incentive Plan Awards |
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||
Name | Grant Date | Approval Date | Award Type | Thres- hold ($)(1) | Target ($)(1) | Maximum ($)(1) | Thres- hold (#)(2) | Target (#)(2) | Maximum (#)(2) | All Other Stock Awards: Number of of Stock or Units (#)(3) | All Other Option Awards: Number of Securities Underlying Options (#)(4) | Exercise or Price of Option Awards ($/Sh)(4) | Grant Date Fair Value of Stock and Option Awards ($)(5) | |||||||||||||||||||||||||||||||||||||
Davis | 3/31/2021 | 2/26/2021 | PSU |
|
|
|
|
|
|
|
|
| 0 | 86,102 | 172,204 |
|
|
|
|
|
|
|
|
| $6,324,576 | |||||||||||||||||||||||||
| 5/4/2021 | 2/26/2021 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 290,272 | 73.73 | 2,760,003 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | $0 | $ | 1,915,274 | $ | 3,830,548 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||
Frazier | 3/31/2021 | 2/26/2021 | PSU |
|
|
|
|
|
|
|
|
| 0 | 100,607 | 201,214 |
|
|
|
|
|
|
|
|
| 7,390,092 | |||||||||||||||||||||||||
| 5/4/2021 | 2/26/2021 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 339,177 | 73.73 | 3,225,004 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 1,894,658 | 3,789,316 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
Litchfield | 3/31/2021 | 2/26/2021 | PSU |
|
|
|
|
|
|
|
|
| 0 | 20,590 | 41,180 |
|
|
|
|
|
|
|
|
| 1,512,420 | |||||||||||||||||||||||||
| 5/4/2021 | 2/26/2021 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 69,413 | 73.73 | 660,001 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 800,137 | 1,600,274 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
Clyburn | 3/31/2021 | 2/26/2021 | PSU |
|
|
|
|
|
|
|
|
| 0 | 40,243 | 80,486 |
|
|
|
|
|
|
|
|
| 2,956,021 | |||||||||||||||||||||||||
| 5/4/2021 | 3/22/2021 | RSU | (6) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 40,690 |
|
|
|
|
|
| 3,000,009 | ||||||||||||||||||||||
| 5/4/2021 | 2/26/2021 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 135,671 | 73.73 | 1,290,003 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 975,342 | 1,950,684 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
DeLuca, Jr. | 3/31/2021 | 2/26/2021 | PSU |
|
|
|
|
|
|
|
|
| 0 | 25,269 | 50,538 |
|
|
|
|
|
|
|
|
| 1,856,135 | |||||||||||||||||||||||||
| 5/4/2021 | 3/22/2021 | RSU |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 27,126 |
|
|
|
|
|
| 1,999,981 | |||||||||||||||||||||||
| 5/4/2021 | 2/26/2021 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 85,188 | 73.73 | 809,999 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 800,000 | 1,600,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
Li | 3/31/2021 | 2/26/2021 | PSU |
|
|
|
|
|
|
|
|
| 0 | 28,077 | 56,154 |
|
|
|
|
|
|
|
|
| 2,062,363 | |||||||||||||||||||||||||
| 5/4/2021 | 2/26/2021 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 94,654 | 73.73 | 900,003 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 946,486 | 1,892,972 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
Estimated Future Payouts |
Estimated Future Payouts | All Other | All Other | Exercise | Grant Date | |||||||||||||||||||||||||||||||||||||||||||||
Name | Grant Date | Approval | Award | Thres- | Target | Maximum | Thres- | Target | Maximum | |||||||||||||||||||||||||||||||||||||||||
Davis | 3/31/23 | 3/27/23 | PSU |
|
|
|
|
|
|
|
|
| 0 | 88,824 | 177,648 |
|
|
|
|
|
|
|
|
| $9,997,585 | |||||||||||||||||||||||||
| 5/2/23 | 1/23/23 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 186,722 | $117.89 | 4,050,000 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | $0 | $ | 2,422,500 | $ | 4,845,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||
Litchfield | 3/31/23 | 3/27/23 | PSU |
|
|
|
|
|
|
|
|
| 0 | 27,963 | 55,926 |
|
|
|
|
|
|
|
|
| 3,147,375 | |||||||||||||||||||||||||
| 5/2/23 | 1/23/23 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 58,783 | 117.89 | 1,275,003 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 1,125,000 | 2,250,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
Chattopadhyay | 3/31/23 | 3/27/23 | PSU |
|
|
|
|
|
|
|
|
| 0 | 21,713 | 43,426 |
|
|
|
|
|
|
|
|
| 2,443,907 | |||||||||||||||||||||||||
| 5/2/23 | 1/23/23 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 45,643 | 117.89 | 989,997 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 941,806 | 1,883,612 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
DeLuca | 3/31/23 | 3/27/23 | PSU |
|
|
|
|
|
|
|
|
| 0 | 21,055 | 42,110 |
|
|
|
|
|
|
|
|
| 2,369,846 | |||||||||||||||||||||||||
| 5/2/23 | 1/23/23 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 44,260 | 117.89 | 959,999 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 925,000 | 1,850,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
Li | 3/31/23 | 3/27/23 | PSU |
|
|
|
|
|
|
|
|
| 0 | 36,846 | 73,692 |
|
|
|
|
|
|
|
|
| 4,147,202 | |||||||||||||||||||||||||
| 5/2/23 | 1/23/23 | Options |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 77,455 | 117.89 | 1,679,999 | |||||||||||||||||||||||||
|
|
|
|
|
|
|
| EIP | 0 | 1,400,000 | 2,800,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
| (1) | Amounts represent awards under the EIP, which equal a specified percentage of base salary as in effect on December 31, |
| (2) | The payout of PSUs can range from zero for below threshold performance to a maximum of 200% of target, depending on the level of achievement of the applicable performance goals. |
| (3) |
|
| (4) | Stock options generally vest and become exercisable in equal installments on the first, second and third anniversaries of the grant date. |
| (5) | This column represents the full grant date fair value of PSUs, RSUs and stock options granted to each of the |
|
Merck & Co., Inc. 2022 2024 Proxy Statement
| 74 | ç ç ç ç |
Grants of Plan-Based Awards Narrative Information Relating to the Grants of Plan-Based Awards Table |
Narrative Information Relating to the Grants of Plan-Based Awards Table
General Information Regarding the EIP
The EIP is a shareholder-approved plan that is administered by the C&MD Committee. It is designed to provide cash awards to employees who are subject to Section 16 of the Securities Exchange Act of 1934, as amended, as follows:
Each executive officer is assigned a target award opportunity that is expressed as a multiple of salary.
The Company performance component (as reflected by the Company Scorecard) is multiplied by the target award opportunity.
The Company performance component can range between 50% and 200% of target.
If the combined results of the three metrics do not total at least 50, no payout will be made.
General Information Regarding Long-Term Incentives
Stock Options
Stock options enable executives to share in the financial gain derived from the potential appreciation in stock price from the date the option is granted until the date the option is exercised. The exercise price of a stock option is set as the closing price of Merck common stock as reported on the NYSE on the grant date (unless a higher grant price is required under local law).date.
Subject to their terms, stock options generally vest and become exercisable in equal installments on the first, second and third anniversaries of the grant date and expire on the day before the tenth anniversary of the grant date.
RSUs
RSUs, subject to their terms, generally vest and become payable in equal installments in shares of Merck common stock on the first, second and third anniversaries of the grant date. Dividend equivalents are accrued and paid out in cash if, and when, the RSUs vest. Mr. Clyburn and Mr. DeLuca, Jr. each received special one-time RSU retention grants on May 4, 2021 that would vest in their entirety on the third anniversary of the grant date, subject to their continued employment. In connection with Mr. Clyburn’s resignation as Executive Vice President and President, Human Health, effective February 1, 2022, Mr. Clyburn’s RSU retention grant was forfeited. Please refer to the Retention Actions section on page 58 for more information.
PSUs
PSUs, subject to their terms, generally vest and become payable in shares of Merck common stock at the end of a three-year performance period provided that minimum performance goals are met. Failure to attain the minimum performance goal results in forfeiture of the shares applicable to the respective award opportunity. PSU awards for continuing executives and performance goals are approved by the C&MD Committee within the first 90 days of the applicable performance cycle.
Similar toFor PSUs granted in 2020,2023, final awards will be determined based on the program design for PSUs granted in 2021 was adjusted as a result of the successful spin-off of our Organon business as follows:following:
33%50% of the award will be determined by the Company’s 2021cumulative EPS versus target.target for the three-year performance period (2023-2025).
67%50% of the award will be determined by the Company’s average annual total shareholder returnTSR (inclusive of reinvested dividends) relative to the median total shareholder returnTSR for our primary peer group for the three-year performance period (2021-2023)(2023-2025).
Payouts can range from zero (for below threshold performance) to a maximum of 200% of target.
Dividend equivalents are accrued and paid in shares if, and when, the PSUs vest, and are only applied to the portion of the award that is earned.
Merck & Co., Inc. 2022 2024 Proxy Statement
| ÷ ÷ ÷ ÷ | 75 |
The following table provides details about each outstanding equity award held by the Named Executive Officers as of December 31, 2021.2023.
Outstanding Equity Awards for Fiscal Year Ended December 31, 20212023
| Option Awards | Stock Awards | Option Awards | Stock Awards | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Name | Number of Securities Underlying Unexercised Options Exercisable (#)(1) | Number of Securities Underlying Unexercised Options Unexercisable (#)(1) | Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) | Grant Date | Option Exercise Price ($) | Vesting Date | Option Expiration Date | Number of or Stock Have Vested (#)(2) | Market Value of Shares or Units Stock Have Not Vested ($)(2)(5) | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Other Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Other Rights That Have Not Vested ($)(5) | Number of | Number of | Equity | Grant | Option | Vesting | Option | Number | Market | Equity | Equity | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Davis | 167,613 |
|
| 5/5/17 | $62.07 | 5/5/18 | 5/4/27 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 143,329 |
|
| 5/4/18 | 56.04 | 5/4/19 | 5/3/28 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 77,578 | 38,792 |
| 5/3/19 | 77.62 | 5/3/20 | 5/2/29 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 41,509 | 83,020 |
| 5/1/20 | 75.36 | 5/1/21 | 4/30/30 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| 290,272 |
| 5/4/21 | 73.73 | 5/4/22 | 5/3/31 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| 75,016 | (3) | $5,749,226 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| 172,204 | (4) | 13,197,715 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Frazier | 567,084 |
|
| 5/9/14 | $56.49 | 5/9/15 | 5/8/24 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Davis | 116,370 |
|
| 05/03/19 | $77.62 | 05/03/20 | 05/02/29 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 765,828 |
|
| 5/1/15 | 58.08 | 5/1/16 | 4/30/25 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 839,941 |
|
| 5/10/16 | 53.06 | 5/10/17 | 5/9/26 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 124,529 |
|
| 05/01/20 | 75.36 | 05/01/21 | 04/30/30 |
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 551,360 |
|
| 5/5/17 | 62.07 | 5/5/18 | 5/4/27 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 490,337 |
|
| 5/4/18 | 56.04 | 5/4/19 | 5/3/28 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 290,926 | 145,464 |
| 5/3/19 | 77.62 | 5/3/20 | 5/2/29 |
|
|
|
|
| 193,512 | 96,760 |
| 05/04/21 | 73.73 | 05/04/22 | 05/03/31 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 163,444 | 326,890 |
| 5/1/20 | 75.36 | 5/1/21 | 4/30/30 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| 339,177 |
| 5/4/21 | 73.73 | 5/4/22 | 5/3/31 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 76,051 | 152,104 |
| 05/03/22 | 87.10 | 05/03/23 | 05/02/32 |
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| 295,378 | (3) | $22,637,770 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| 201,214 | (4) | 15,421,041 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| 186,722 |
| 05/02/23 | 117.89 | 05/02/24 | 05/01/33 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| 200,488 | (3) | $21,857,202 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| 177,648 | (4) | 19,367,185 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Litchfield | 38,291 |
|
| 5/1/15 | $58.08 | 5/1/16 | 4/30/25 |
|
|
|
|
| 38,291 |
|
| 05/01/15 | 58.08 | 05/01/16 | 04/30/25 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 41,997 |
|
| 5/10/16 | 53.06 | 5/10/17 | 5/9/26 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 26,465 |
|
| 5/5/17 | 62.07 | 5/5/18 | 5/4/27 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 41,997 |
|
| 05/10/16 | 53.06 | 05/10/17 | 05/09/26 |
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 22,630 |
|
| 5/4/18 | 56.04 | 5/4/19 | 5/3/28 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 11,634 | 5,821 |
| 5/3/19 | 77.62 | 5/3/20 | 5/2/29 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6,770 | 13,543 |
| 5/1/20 | 75.36 | 5/1/21 | 4/30/30 |
|
|
|
|
| 26,465 |
|
| 05/05/17 | 62.07 | 05/05/18 | 05/04/27 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| 69,413 |
| 5/4/21 | 73.73 | 5/4/22 | 5/3/31 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
| 773 | $59,243 |
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 22,630 |
|
| 05/04/18 | 56.04 | 05/04/19 | 05/03/28 |
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
| 1,732 | 132,740 |
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| 6,992 | (3) | $535,867 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| 41,180 | (4) | 3,156,035 | 17,455 |
|
| 05/03/19 | 77.62 | 05/03/20 | 05/02/29 |
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 20,313 |
|
| 05/01/20 | 75.36 | 05/01/21 | 04/30/30 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 46,274 | 23,139 |
| 05/04/21 | 73.73 | 05/04/22 | 05/03/31 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 17,799 | 35,599 |
| 05/03/22 | 87.10 | 05/03/23 | 05/02/32 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| 58,783 |
| 05/02/23 | 117.89 | 05/02/24 | 05/01/33 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| 46,922 | (3) | 5,115,436 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| 55,926 | (4) | 6,097,053 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | 66,913 |
|
| 05/03/19 | 77.62 | 05/03/20 | 05/02/29 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 87,170 |
|
| 05/01/20 | 75.36 | 05/01/21 | 04/30/30 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 58,894 | 29,449 |
| 05/04/21 | 73.73 | 05/04/22 | 05/03/31 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 19,417 | 38,835 |
| 05/03/22 | 87.10 | 05/03/23 | 05/02/32 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| 45,643 |
| 05/02/23 | 117.89 | 05/02/24 | 05/01/33 |
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| 51,188 | (3) | 5,580,516 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| 43,426 | (4) | 4,734,303 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Merck & Co., Inc. 2022 2024 Proxy Statement
| 76 | ç ç ç ç |
Outstanding Equity Awards |
Outstanding Equity Awards for Fiscal Year Ended December 31, 20212023
| Option Awards | Stock Awards | Option Awards | Stock Awards | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Name | Number of Securities Underlying Unexercised Options Exercisable (#)(1) | Number of Securities Underlying Unexercised Options Unexercisable (#)(1) | Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) | Grant Date | Option Exercise Price ($) | Vesting Date | Option Expiration Date | Number of Shares or Units of Stock That Have Not Vested (#)(2) | Market Value of Shares or Units of Stock That Have Not Vested ($)(2)(5) | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Other Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Other Rights That Have Not Vested ($)(5) | Number of | Number of | Equity | Grant | Option | Vesting | Option | Number | Market | Equity | Equity | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Clyburn | 19,848 | 5/2/14 | $56.49 | 5/2/15 | 5/1/24 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DeLuca | 58,185 | 05/03/19 | $77.62 | 05/03/20 | 05/02/29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 5,668 | 2/9/15 | 56.60 | 2/9/16 | 2/8/25 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 44,673 | 5/1/15 | 58.08 | 5/1/16 | 4/30/25 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 77,830 | 05/01/20 | 75.36 | 05/01/21 | 04/30/30 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 55,995 | 5/10/16 | 53.06 | 5/10/17 | 5/9/26 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 35,287 | 5/5/17 | 62.07 | 5/5/18 | 5/4/27 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 41,489 | 5/4/18 | 56.04 | 5/4/19 | 5/3/28 | 56,792 | 28,396 | 05/04/21 | 73.73 | 05/04/22 | 05/03/31 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 34,910 | 17,456 | 5/3/19 | 80.00 | 5/3/20 | 5/2/29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 25,942 | 51,888 | 5/1/20 | 77.67 | 5/1/21 | 4/30/30 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 19,417 | 38,835 | 05/03/22 | 87.10 | 05/03/23 | 05/02/32 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 135,671 | 5/4/21 | 73.73 | 5/4/22 | 5/3/31 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 40,690 | $3,118,482 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 46,886 | (3) | $3,593,343 | 44,260 | 05/02/23 | 117.89 | 05/02/24 | 05/01/33 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 80,486 | (4) | 6,168,447 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DeLuca, Jr. | 59,564 | 5/1/15 | 58.08 | 5/1/16 | 4/30/25 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 104,993 | 5/10/16 | 53.06 | 5/10/17 | 5/9/26 | 27,126 | $2,957,277 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 66,163 | 5/5/17 | 62.07 | 5/5/18 | 5/4/27 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 75,436 | 5/4/18 | 56.04 | 5/4/19 | 5/3/28 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 51,188 | (3) | $5,580,516 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 38,790 | 19,395 | 5/3/19 | 80.00 | 5/3/20 | 5/2/29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 25,942 | 51,888 | 5/1/20 | 77.67 | 5/1/21 | 4/30/30 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 85,188 | 5/4/21 | 73.73 | 5/4/22 | 5/3/31 |
|
|
|
|
|
|
|
|
|
| 42,110 | (4) | 4,590,832 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 27,126 | $2,078,937 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 46,886 | (3) | $3,593,343 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 50,538 | (4) | 3,873,232 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Li | 14,702 | 5/5/17 | 62.07 | 5/5/18 | 5/4/27 | 14,702 | 05/05/17 | 62.07 | 05/05/18 | 05/04/27 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15,087 | 5/4/18 | 56.04 | 5/4/19 | 5/3/28 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 11,634 | 5,821 | 5/3/19 | 80.00 | 5/3/20 | 5/2/29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15,087 | 05/04/18 | 56.04 | 05/04/19 | 05/03/28 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 7,004 | 14,010 | 5/1/20 | 77.67 | 5/1/21 | 4/30/30 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1,296 | 2,595 | 5/1/20 | 77.67 | 5/1/21 | 4/30/30 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 94,654 | 5/4/21 | 73.73 | 5/4/22 | 5/3/31 | 17,455 | 05/03/19 | 77.62 | 05/03/20 | 05/02/29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 773 | $59,243 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1,791 | 137,262 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 21,014 | 05/01/20 | 75.36 | 05/01/21 | 04/30/30 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 332 | 25,444 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 8,574 | (3) | $657,111 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 56,154 | (4) | 4,303,643 | 3,891 | 05/01/20 | 75.36 | 05/01/21 | 04/30/30 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 63,100 | 31,554 | 05/04/21 | 73.73 | 05/04/22 | 05/03/31 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 25,242 | 50,486 | 05/03/22 | 87.10 | 05/03/23 | 05/02/32 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 77,455 | 05/02/23 | 117.89 | 05/02/24 | 05/01/33 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 66,544 | (3) | 7,254,627 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| 73,692 | (4) | 8,033,902 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (1) | Stock options generally vest and become exercisable in equal installments on the first, second and third anniversaries of the grant date, and expire on the day before the tenth anniversary of the grant date. The date set forth in the “Vesting Date” column represents the first vesting date for such award. |
Merck & Co., Inc. 2022 Proxy Statement
|
| (2) | The grant and vesting dates of the unvested RSU award in this column are as follows for Mr. DeLuca: the grant date of the 27,126 RSUs is May 4, 2021. This was a special one-time RSU retention grant that is scheduled to vest in its entirety on May 4, 2024, subject to Mr. DeLuca’s continued employment. The RSUs are payable in shares of Merck common |
| (3) | Maximum (200% of target) of PSUs granted during |
| (4) | Maximum (200% of target) of PSUs granted during |
| (5) | The market value of the units reported in this column was computed by multiplying the number of such units by |
Merck & Co., Inc. 2022 2024 Proxy Statement
|
÷ ÷ ÷ | 77 |
Option Exercises and Stock Vested
The following table provides information about stock options that were exercised and stock units that vested during 2021.2023.
Option Exercises and Stock Vested for Fiscal Year Ended December 31, 20212023
| Option Awards | Stock Awards | Option Awards | Stock Awards | |||||||||||||||||||||||||||||
Name | Number of Shares Acquired on Exercise (#) | Value Realized on Exercise ($)(1) | Number of Shares Acquired on Vesting (#)(2) | Value Realized on Vesting ($)(3) | Number of Shares Acquired on Exercise (#) | Value Realized on Exercise ($)(1) | Number of Shares Acquired on Vesting (#)(2) | Value Realized on Vesting ($)(3) | ||||||||||||||||||||||||
Davis | — | $— | 53,457 | (a) | $3,929,624 | |||||||||||||||||||||||||||
Frazier | 663,881 | 25,632,956 | 200,456 | (a) | 14,735,521 | |||||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||
Davis | 143,329 | $8,440,688 | 148,374 | (a) | $17,720,307 | |||||||||||||||||||||||||||
Litchfield | 22,683 | 782,813 | 4,584 | (a) | 336,970 | |||||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||||||||||
Litchfield | — | — | 36,347 | (a)(b) | 4,337,493 | |||||||||||||||||||||||||||
Clyburn | 59,086 | 2,173,380 | 24,056 | (a) | 1,768,357 | |||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||
Chattopadhyay | 218,049 | 12,488,854 | 45,157 | (a) | 5,393,101 | |||||||||||||||||||||||||||
DeLuca, Jr. | 165,187 | 6,107,450 | 26,728 | (a) | 1,964,775 | |||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||
DeLuca | 141,599 | 8,027,422 | 43,544 | (a) | 5,200,460 | |||||||||||||||||||||||||||
Li | — | — | 4,584 | (a) | 336,970 | |||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||
Li | — | — | 49,446 | (a)(b) | 5,901,130 | |||||||||||||||||||||||||||
| (1) | This column represents the values realized upon stock option exercises during |
| (2) | This column represents the vesting during |
| (a) | PSUs granted in |
| (b) | In addition to the 2021 PSU grant, Ms. Litchfield and Dr. Li also had RSUs that were granted on May 1, 2020, prior to them becoming Section 16 Officers, and that partially vested in 2023. These RSUs vested in equal installments on the first, second and third anniversaries of the grant date. The number of RSUs that vested for Ms. Litchfield |
| (3) | The value realized for PSUs was determined by multiplying the number of vested units by the market price of Merck common stock on |
Merck & Co., Inc. 2022 2024 Proxy Statement
| 78 | ||||||||||
ç ç ç ç |
|
The table below sets forthprovides information concerning the present value of benefits accumulated by the Named Executive Officers from:from the Merck U.S. Pension Plan (the “Qualified Plan”), the MSD Supplemental Retirement Plan (the “SRP”), and U.K. Pension Plan (see footnote 4 below). The terms of the U.S. plansQualified Plan and SRP are described below.
Pension Benefits for Fiscal Year Ended December 31, 20212023
Name | Plan Name | Number of Years Credited Service (#)(1) | Number of Years Cash Balance Service (#)(2) | Present Value of Accumulated Benefit ($)(3) | Payments During Last Fiscal Year ($) | Plan Name | Number of Years Credited Service (#)(1) | Number of Years Cash Balance Service (#)(2) | Present Value of Accumulated Benefit ($)(3) | Payments During Last Fiscal Year ($) | ||||||||||||||||||||||||||||||
Davis |
| Qualified Plan |
|
| — |
|
| 7.67 |
|
| $211,168 |
|
| $0 |
| |||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||||||||||
Davis | Qualified Plan | — | 9.67 | $266,289 | $0 | |||||||||||||||||||||||||||||||||||
|
|
| SRP |
|
| — |
|
| 7.67 |
|
| 1,465,456 |
|
| 0 |
| |||||||||||||||||||||||||
Frazier |
| Qualified Plan |
|
| 27.50 |
|
| 29.58 |
|
| 1,608,271 |
|
| 0 |
| |||||||||||||||||||||||||
|
|
| SRP |
|
| 27.50 |
|
| 29.58 |
|
| 29,403,403 |
|
| 0 |
| SRP | — | 9.67 | 2,241,757 | 0 | ||||||||||||||||||||
Litchfield(4) | ||||||||||||||||||||||||||||||||||||||||
Litchfield(4) | ||||||||||||||||||||||||||||||||||||||||
Litchfield(4) | ||||||||||||||||||||||||||||||||||||||||
Litchfield(4) |
| Qualified Plan |
|
| — |
|
| 31.25 |
|
| 75,094 |
|
| 0 |
| Qualified Plan | — | 33.25 | 141,103 | 0 | ||||||||||||||||||||
|
| SRP |
|
| — |
|
| 31.25 |
|
| 221,036 |
|
| 0 |
| |||||||||||||||||||||||||
|
|
| U.K. Pension Plan |
|
| 26.25 |
|
| — |
|
| 4,663,183 |
|
| 0 |
| |||||||||||||||||||||||||
| SRP | — | 33.25 | 663,386 | 0 | ||||||||||||||||||||||||||||||||||||
Clyburn |
| Qualified Plan |
|
| 11.50 |
|
| 13.92 |
|
| 687,878 |
|
| 0 |
| |||||||||||||||||||||||||
|
|
| SRP |
|
| 11.50 |
|
| 13.92 |
|
| 2,821,094 |
|
| 0 |
| |||||||||||||||||||||||||
DeLuca, Jr. |
| Qualified Plan |
|
| 8.00 |
|
| 10.25 |
|
| 513,036 |
|
| 0 |
| |||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
| U.K. Pension Plan | 26.25 | — | 2,646,601 | 0 | |||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | Qualified Plan | 10.00 | 14.08 | 621,463 | 0 | |||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
| SRP | 10.00 | 14.08 | 2,847,348 | 0 | |||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||
DeLuca | Qualified Plan | 8.00 | 12.25 | 518,902 | 0 | |||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
|
| SRP |
|
| 8.00 |
|
| 10.25 |
|
| 2,350,605 |
|
| 0 |
| SRP | 8.00 | 12.25 | 2,484,870 | 0 | ||||||||||||||||||||
Li |
| Qualified Plan |
|
| — |
|
| 4.75 |
|
| 126,298 |
|
| 0 |
| |||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||||||||||
Li | Qualified Plan | — | 6.75 | 192,053 | 0 | |||||||||||||||||||||||||||||||||||
|
|
| SRP |
|
| — |
|
| 4.75 |
|
| 294,500 |
|
| 0 |
| |||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||||||||||||
|
| SRP | — | 6.75 | 778,198 | 0 | |||||||||||||||||||||||||||||||||||
| (1) | This column shows the number of years of Credited Service that is used for benefit accrual purposes and eligibility purposes under the Final Average Pay formula of the Qualified Plan and the SRP. The Final Average Pay formula is applicable only for participants who were actively employed on December 31, 2012. Participants hired (or rehired) after December 31, 2012 receive benefits under a Cash Balance formula that does not rely on Credited Service. |
For employees actively employed on December 31, 2012, Credited Service for the Final Average Pay formula begins with the January 1 or July 1 that coincides with or follows a participant’s hire date and ends with the last full month of employment. Credited Service is earned through the earlier of termination or December 31, 2019. After December 31, 2019, all benefits will be calculated under the Cash Balance formula. A maximum of 35 years of Credited Service may be earned. Mr. Davis, Ms. Litchfield and Dr. Li do not have Credited Service because they entered the |
The number of years of Credited Service for Ms. Litchfield in the U.K. Pension Plan row shown is used for benefit accrual purposes while she participated in the plan prior to her transfer from the U.K. to the U.S. |
| (2) | This column shows the number of years of Cash Balance Service that is used for benefit accrual purposes under the Cash Balance formula of the Qualified Plan and the SRP. |
Cash Balance Service begins on a participant’s first day of employment, includes all years and completed months of service, and ends on the participant’s date of termination of employment. |
The Cash Balance Service for Ms. Litchfield is based on her original hire date and is used to determine the pay credit level under the Cash Balance formula. Benefit accruals under the Cash Balance formula did not start until January 1, 2020, effective with her transfer to the U.S. Prior to January 1, 2020, Ms. Litchfield was not a U.S. based employee and, therefore, not eligible for the |
| (3) | For the Qualified Plan and the SRP, the actuarial present value is calculated using the same assumptions used for financial statement reporting purposes as set forth in the footnotes to our financial statements, except that commencement is assumed at the earliest unreduced retirement age (with no pre-retirement mortality). The earliest unreduced retirement age is the earlier of age 62 and 10 years of Credited Service (including service under the Cash Balance formula) or age 65 with no service requirement. |
Discount rate equals 3.0%5.25% for the Qualified Plan and 2.8%5.38% for the SRP;
Merck & Co., Inc. 2022 Proxy Statement
|
|
Interest crediting rate equals 4.95%5.35% for both the Qualified Plan and the SRP;
Mortality based on 100% of the sex distinct Pri-2012 White Collar Mortality Table, with projection based on modified MP-2021 Projection Scale using a 0.75% ultimate rate at most ages;
Merck & Co., Inc. 2024 Proxy Statement
Pension Benefits | ÷ ÷ ÷ ÷ | 79 |
| • | Mortality based on 100% of the sex distinct Pri-2012 White Collar Mortality Table, with projection based on modified MP-2021 Projection Scale using a 0.75% ultimate rate at most ages; |
Future lump sum conversion factors calculated by implied forward rates embedded in the 12/31/20212023 Willis Towers Watson RATE:Link 60th to 90th percentile yield curve and mortality defined in Internal Revenue Code Section 417(e)(3); and
Assumes that 80% of retirees elect a lump sum and the remaining 20% elect an annuity for the Qualified Plan and assumes 100% of retirees elect a lump sum for the SRP.
| (4) | The amounts in the table for the U.K. Pension Plan for Ms. Litchfield reflect benefits accrued during her participation in the plan plus legally required U.K. pension consumer price index increases. The amounts reported represent the actuarial present value of the accrued benefit payable at age 65 and converted from GBP to USD using an exchange rate ($1/£) of |
The NEOs participate in both U.S. defined benefit plans, as do other U.S.-based Merck Sharp & Dohme Corp.LLC salaried employees. Benefits payable under the Qualified Plan and the SRP are based on several formulas.
Beginning in 2013, a Cash Balance formula was added to replace the Final Average Pay (“FAP”) formula. Employees eligible for U.S. benefits on December 31, 2012 receive transition benefits, which provide the greater of the benefit under the Cash Balance and FAP formulas through December 31, 2019, or the date the participant terminates employment or loses retirement plan eligibility, if earlier. Only Mr. Frazier, Mr. DeLuca Jr. and Mr. ClyburnChattopadhyay are eligible for transition benefits.
Final Average Pay Formula: For service prior to January 1, 2013, benefits are calculated and shown as a single life annuity normally payable at age 65 (normal retirement date or “NRD”). The amount equals:

* Limited to 31.25
Final Average Pay. The average of a participant’s highest five consecutive calendar years of Total Pay for the 10 years before the earlier of:
Termination of employment, or
December 31, 2019, if eligible for the transition provisions.
Cash Balance Formula: For service starting January 1, 2013, benefits are calculated and shown as an account balance that grows with annual pay credits according to the following schedule:
Age + Cash Balance Service at 12/31 | Percent of Total Pay credited to Account Balance | |||
| At Least | Less Than | |||
| — | 40 | 4.5% | ||
| 40 | 50 | 5.5% | ||
| 50 | 60 | 6.5% | ||
| 60 | 70 | 8.0% | ||
| 70 | — | 10.0% | ||
The account balance also earns interest credits every year at the annual rate of the change in the Consumer Price Index plus 3% (and not less than 3.3%). The account balance is the sum of annual pay credits and interest credits.
Final Average Pay. The average of a participant’s highest five consecutive calendar years of Total Pay for the 10 years before the earlier of:
Termination of employment, or
December 31, 2019, if eligible for the transition provisions.
Total Pay is generally base salary and EIP for both the FAP and Cash Balance formulas for the NEOs.
Vesting. A participant is generally vested after three years of vesting service. All NEOs are vested. A participant who is vested and terminates employment can commence receiving a reduced pension benefit after attaining age 55. FAP benefits are reduced on an actuarial basis. Participants who only have vested benefits under the Cash Balance formula can commence payment of their Qualified Plan benefit immediately upon termination.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 80 | ||||||||||
ç ç ç ç |
Pension Benefits |
Early Retirement Subsidies. Under the FAP formula, a participant who is age 55 with at least 10 years of credited service is entitled to early retirement subsidies. Under this provision, unreduced benefits may begin at age 62 and benefits that begin before 62 are only reduced by 3% per year. As of December 31, 2023, Mr. Chattopadhyay, and Mr. DeLuca are retirement eligible under the Qualified Plan and the SRP (i.e., at least age 55 and having completed at least 10 years of Credited Service).
SRP Benefits. The Qualified Plan benefits are limited by the Internal Revenue Code. The SRP is an unfunded plan maintained to provide benefits according to the formulas described above without regard to those limits. The SRP also may include benefits based on compensation deferred into the Merck Deferral Program.
Forms of Benefit. In the Qualified Plan and in the SRP for accruals prior to 2005, a participant generally can choose from several annuity options or a lump sum. SRP accruals post-2004 are payable in a lump sum or installments of 5 to 10 years. All forms of benefit are actuarially equivalent to the single life annuity.
Merck & Co., Inc. 2022 2024 Proxy Statement
|
÷ ÷ ÷ | 81 |
Nonqualified Deferred Compensation
The following table shows the executive contributions, earnings, and account balances for the Named Executive Officers in the Merck Deferral Program, an unfunded, nonqualified, unsecured deferred compensation plan. The Merck Deferral Program allows participants who are executive officers to defer all or a portion of annual bonus (but not less than $3,000), and/or up to 50% of base salary, subject to certain limitations.
Nonqualified Deferred Compensation for Fiscal Year Ended December 31, 20212023
Name | Executive in 2021 ($) | Registrant in 2021 ($)(1) | Aggregate Earnings in 2021 ($)(2) | Aggregate Withdrawals/ in 2021 ($) | Aggregate Balance at 12/31/21 ($)(3) | Executive Contributions in 2023 ($) | Registrant Contributions in 2023 ($)(1) | Aggregate Earnings in 2023 ($)(2) | Aggregate Withdrawals/ Distributions in 2023 ($) | Aggregate Balance at 12/31/23 ($)(3) | ||||||||||||||||||||||||||||||
Davis | $0 | $92,124 | $139,997 | $0 | $1,077,132 | |||||||||||||||||||||||||||||||||||
Frazier | 0 | 153,002 | 2,454,256 | 0 | 25,865,809 | |||||||||||||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||||||||||
Davis | ||||||||||||||||||||||||||||||||||||||||
Davis | $0 | $242,730 | $213,020 | $0 | $1,517,885 | |||||||||||||||||||||||||||||||||||
Litchfield | 0 | 36,720 | 9,911 | 0 | 93,968 | |||||||||||||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||||||||||||||||||
Litchfield | ||||||||||||||||||||||||||||||||||||||||
Litchfield | 0 | 112,315 | 42,976 | 0 | 310,519 | |||||||||||||||||||||||||||||||||||
Clyburn | 38,758 | 59,176 | 221,064 | 0 | 1,748,791 | |||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | ||||||||||||||||||||||||||||||||||||||||
Chattopadhyay | 0 | 99,300 | 700,674 | 0 | 11,068,547 | |||||||||||||||||||||||||||||||||||
DeLuca, Jr. | 166,933 | 48,857 | 669,716 | 0 | 4,135,862 | |||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||
DeLuca | ||||||||||||||||||||||||||||||||||||||||
DeLuca | 226,950 | 94,081 | 937,585 | 0 | 4,877,634 | |||||||||||||||||||||||||||||||||||
Li | 0 | 43,007 | 44,987 | 0 | 227,197 | |||||||||||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||||||||||
Li | ||||||||||||||||||||||||||||||||||||||||
Li | 0 | 146,717 | 100,553 | 0 | 525,693 | |||||||||||||||||||||||||||||||||||
| (1) | The amounts disclosed in this column represent the Company’s annual 4.5% credit of eligible pay in excess of the IRS limit to the NEOs’ accounts under the Merck Deferral Program. These amounts are included within the amount disclosed in the “All Other Compensation” column of the Summary Compensation table for each applicable NEO for |
| (2) | This column represents dividends earned plus changes in market and account value (investment earnings or losses) for the period of January 1, |
| (3) | This column includes deferred compensation earned in earlier years which was disclosed as “Salary,” “Non-Equity Incentive Plan Compensation” or “All Other Compensation” in the Summary Compensation table of prior proxy statements as follows: Mr. Davis, |
Merck Deferral Program Investments. Account balances may be invested in phantom investments selected by the executive from an array of investment options that mirrors the funds in the Merck U.S. Savings Plan.
Distributions. When participants elect to defer amounts into the Merck Deferral Program, they also elect when the amounts ultimately will be distributed to them. Distributions may either be made in a specific year (regardless of whether employment has then ended) or at a time that begins at or after the executive’s employment has ended. Distributions can be made in a lump sum or up to 15 annual installments. Distributions from the Merck common stock fund are made in shares, with cash payable for any partial share.
Merck & Co., Inc. 2022 2024 Proxy Statement
| 82 | ||||||||||
ç ç ç ç |
|
Termination or a Change in Control
The section below describes the payments and benefits that may be made to the NEOs upon separation, either pursuant to individual agreements ortermination from employment, including in connection with a change in control (as defined below). For payments that may be made to a participantthe NEOs upon a retirement, other than in connection with a separation or change in control, see the Pension Benefits table and related narrative beginning on page 72.78.
Separation
The Company provides separation pay and benefits to all of its U.S.-based employees who are employees of Merck Sharp & Dohme Corp., including the NEOs, pursuant to the Merck & Co., Inc. U.S. Separation Benefits Plan, as amended (the “Separation Plan”). An amount related to the executive’s target annual incentive award is provided under certain circumstances. To be eligible for all of the benefits described below, a general release of claims is required, as well as compliance with non-disparagement, cooperation with litigation and, in some cases, non-competition and non-solicitation agreements in connection with, and at the time of, the separation.
Severance Pay.The Separation Plan provides severance pay and benefits to certain eligible employees, including the NEOs, whose employment is terminated due to organizational changes, including discontinuance of operations, location closings, corporate restructuring, or a general reduction in work force.force reduction. For eligible separations during 2021, certain management-level employees, including the NEOs, were eligible to receive the following severance pay and benefits are payable in a lump sum.under the Separation Plan:
A cash lump sum severance payment, the amount which for the NEOs, is determined as follows:
Years of Continuous Service at Separation Date | ||
Less than 1 year | 26 | |
1-4 years | 40 | |
5 years or more | 40 plus 2 additional weeks for each year of continuous service beyond 4 years (maximum 78 total) | |
Health and Welfare Continuation. Separated employees are eligible for continued
Continued participation in the Company’s medical, dental, and basic life insurance plans at active-employee rates for 26 to 78 weeks depending(determined based on their years of continuous service, by paying contributions at the same rate as paid by active employees.service); and
Outplacement Assistance. Certain management-level employees, including the NEOs, are eligibleservices for up to 12 monthsmonths.
To be eligible for the severance pay and benefits described above, a general release of executive outplacement services.claims is required, as well as compliance with applicable restrictive covenants.
EIPEffects of Termination on Other Awards or Under Other Benefit Plans
As part of our standard practiceThe NEOs may be eligible for separated employeesadditional payments and depending on the date of separation, we may pay an amount in lieu of a bonus payout under the EIP.benefits upon termination from employment as described below.
| • | EIP Awards. The NEOs may be eligible for payments in lieu of EIP bonus payouts as follows: |
If employment terminates following the end of the performance year, the executive will be considered for a bonus on the same terms and conditions as other employees with respect to the previous year’s performance.
Timing of Termination | Eligibility for Pay in Lieu of EIP | Determination of Amount of Pay in Lieu of EIP | ||
Termination occurs following the end of the performance year | Eligibility determined in accordance with same terms and conditions as other employees participating in the EIP | Amount determined in accordance with same terms and conditions as other employees participating in the EIP | ||
Termination occurs between January 1 and June 30 of the performance year | Eligible for special payment if NEO is retirement eligible | Amount of the special payment is based on the NEO’s target award and the number of months worked in the performance year | ||
Termination occurs after June 30 and before December 31 of the performance year | Eligible for special payment | Amount of the special payment is based on the NEO’s target award and the number of months worked in the performance year | ||
If employment terminates between January 1 and June 30 of the performance year, the employee is not eligible for payment of a bonus for the year in which separation occurred, unless the employee is retirement eligible. Retirement eligible employees may receive a special payment in lieu of any award under the EIP. The amount of the special payment is based on his or her target award and the number of months worked in the current year.
If employment terminates after June 30 and before December 31 of the performance year, a special payment is made in lieu of any award under the EIP. The amount of the special payment is based on the employee’s target award and the number of months worked in the current year.
| • | Retirement Plan Bridge. NEOs who are age 50 or above with at least 10 years of Cash Balance Service as of December 31 of the year of separation may be eligible for a pro-rata portion of the early retirement subsidies described in the Pension Benefits section beginning on page 78. The pro-rata portion equals the percentage of the employee’s Credited Service on the separation date divided by the Credited Service the employee would have had if employment had continued until the employee was first eligible to be treated as an early retiree. This benefit is only provided in exchange for a valid release of claims. |
Merck & Co., Inc. 2022 2024 Proxy Statement
Potential Payments Upon Termination or a Change in Control
Merck & Co., Inc. U.S. Separation Benefits Plan, as amended (the “Separation Plan”) |
÷ ÷ ÷ | 83 |
| • | Retiree Healthcare Bridge. NEOs who are age 50 or above with 10 years of Cash Balance Service as of December 31 of the year of separation may be eligible for subsidized retiree medical benefits in accordance with the plan provisions applicable to similarly situated retired employees, as may be amended from time to time. This benefit is only provided in exchange for a valid release of claims. |
Options, RSUs and PSUs
| • | Upon retirement: |
| • | Options: If an NEO retiree has unvested stock options, the portion of the applicable stock option that would have become vested and exercisable according to its original schedule within one year of the retirement date will vest and become exercisable on its applicable vesting date and the remainder of the stock option will expire immediately. All vested stock options (including those that become vested in connection with the NEO’s retirement) will expire on the earlier of (a) the fifth anniversary of the NEO’s retirement date or (b) the original expiration date of the stock options. |
Effects Under Other Benefit Plans
| • | RSUs: If an NEO retiree has unvested RSUs, a pro rata portion of the unvested RSUs will vest and the underlying shares of Merck common stock and the applicable accrued dividend equivalents will be distributed to the retiree on the next scheduled vesting date following the retirement date. The pro rata portion is determined based on the number of completed months of employment during the three-year vesting period relative to the total length of the period, i.e., 36 months, less any RSUs that had already vested under the applicable award. The remaining unvested portion of the award is forfeited as of the retirement date. |
Separated employees may be eligible for additional benefits under other plans as described below. In general, these benefits are only provided in exchange for a valid release of claims.
| • | PSUs: On the regularly scheduled determination date set forth in the applicable award agreement, an NEO retiree will become vested in a pro-rata portion of the PSUs that become eligible to vest based on the actual attainment level of the applicable performance metrics, if any, and the underlying shares of Merck common stock and applicable accrued dividend equivalents will be distributed on the original settlement date set forth in the applicable award agreement. The pro rata portion is determined based on the number of completed months of employment during the three-year performance period relative to the total length of the period, i.e., 36 months. The remaining unearned portion of the award is forfeited as of the retirement date. |
Retirement Plan Bridge. This benefit is available to employees who would have been at least age 50 with at least 10 years of Cash Balance Service as of December 31 of the year in which their separation occurs. These employees receive a pro-rata portion of the enhancement provided by early retirement subsidies as described in the Pension Benefits section beginning on page 72. The pro-rata portion equals the percentage of the employee’s Credited Service on the separation date divided by the Credited Service the employee would have had if employment had continued until the employee was first eligible to be treated as an early retiree.
| • | Upon involuntary termination (other than in the case of a termination by reason of non-performance of duties, sale, death, disability, or gross misconduct): |
Retiree Healthcare Bridge. If the employee is at least age 52 with 10 years of Cash Balance Service as of December 31 of the year in which separation occurs they are eligible for subsidized retiree medical benefits in accordance with the plan provisions applicable to similarly situated retired employees, as they may be amended from time to time.
| • | Options: Unvested stock options expire on the termination date. Vested stock options as of the termination date may be exercised for one year following the NEO’s termination (but not beyond the original term of the stock option). |
Options, RSUs and PSUs. In 2021, all separated employees
| • | RSUs: If the NEO has unvested RSUs, a pro rata portion of the unvested RSUs will vest and the underlying shares of Merck common stock and the applicable accrued dividend equivalents will be distributed to the NEO on the next scheduled vesting date following the NEO’s termination, so long as such termination occurs on or after the first anniversary of the grant date. The pro rata portion is determined based on the number of completed months of employment during the three-year vesting period relative to the total length of the period, i.e., 36 months, less any RSUs that had already vested under the applicable award. The remaining unvested portion of the award is forfeited as of the termination date. |
| • | PSUs: On the regularly scheduled determination date as set forth in the applicable award agreement, the NEO will become vested in a pro-rata portion of the PSUs that become eligible to vest based on the actual attainment level of the applicable performance metrics, if any, so long as such termination occurs on or after the first anniversary of the first day of the applicable performance period, and the underlying shares of Merck common stock and the applicable accrued dividend equivalents will be distributed on the original settlement date set forth in the applicable award agreement. The pro rata portion is determined based on the number of completed months of employment during the three-year performance period relative to the total length of the period, i.e., 36 months. The remaining unearned portion of the award is forfeited as of the termination date. |
NEOs who are entitled to the Retirement Plan Bridge, as described above, are also eligible to be treated in accordance with the equity plan’s provisions applicable to retired employees with respect to stock options granted to them before 2013. For stock option, RSU and PSU grants occurring during or after 2013, generally, separated “bridged” employees are eligible to be treated in accordance with the equity plan’s provisions applicable to retired employees only if they are also eligible for subsidized retiree medical benefits. If theythe “bridged” employees are not also eligible for subsidized retiree medical benefits, separated bridged employees will be treated in accordance with the equity plan’s provisions applicable to involuntarily terminated employees.employees, as described above.
Merck & Co., Inc. 2024 Proxy Statement
| 84 | ç ç ç ç | Potential Payments Upon Termination or a Change in Control Individual Agreements and Arrangements
|
Individual Agreements and Arrangements
When Robert M. Davis was appointed Executive Vice President and Chief Financial Officer by the Board, effective April 23, 2014, to compensate Mr. Davis for pension benefits he forfeited upon leaving his prior employer, his offer letter provides for a cash payment of $2,000,000 within 90 days of his termination of employment (other than for cause) provided that he was employed for at least 10 years with no breaks in service. The terms of this offer letter continue in full force and effect following his promotions that occurred in 2021 and 2022.
Effects of Change in Control under Change in Control, Equity, and Other Benefit Plans
Merck & Co., Inc. Change in Control Separation Benefits Plan, as amended (the “Change in Control Plan”)
The Change in Control Plan provides for the following severance payments and benefits upon a termination without “cause,” or a resignation by the NEO for “good reason,” in each case within two years following a change in control:
Merck & Co., Inc. 2022 Proxy Statement
|
Change in Control
Participants in the Change in Control Plan include the NEOs as well as certain other senior executives whose participation was allowed to continue when the C&MD Committee otherwise reduced the size of the participant population in 2012. With respect to the NEOs, the severance benefits described below would be provided upon qualifying terminations of employment within two years following a change in control.
Cash severance paid in a lump sum within 90 days following employment termination in an amount equal to three (for the CEO) or two (for the other NEOs) times the sum of the NEO’s base salary, plus the lesser of (a) the NEO’s target bonus amount or (b) the average of the actual bonuses paid to the NEO in the three years immediately preceding termination while the NEO was serving in the same position as he or she was serving immediately prior to termination, annualized for partial or incomplete years in such position.
Cash severance paid in a lump sum within 90 days following employment termination in an amount equal to three (for the CEO) or two (for the other NEOs) times the sum of (x) the NEO’s base salary, plus (y) the lesser of (a) the NEO’s target bonus amount or (b) the average of the actual bonuses paid to the NEO in the three years immediately preceding termination while the NEO was serving in the same position as he or she was serving immediately prior to termination, annualized for partial or incomplete years in such position;
| • | Pro-rata annual cash incentive at target levels, paid in a lump sum within 90 days following employment |
Continued medical, dental and life insurance benefits at active-employee rates for a period of up to three years for the CEO and for up to two years for the other NEOs, which are reduced by benefits obtained from a subsequent employer;
If the NEO would have attained specified age and service levels within two years following the change in control, then the NEO is entitled to (a) subsidized and/ or unreduced pension benefits upon commencement of pension benefits in accordance with plan terms after termination of employment, and (b) subsidized retiree medical benefits as a retiree under our health plans commencing immediately after the expiration of the benefit continuation period at active-employee rates as described above;
Continued medical, dental and life insurance benefits at active-employee rates for a period of up to three years for the CEO and for up to two years for the other NEOs. These benefits are reduced by benefits obtained from a subsequent employer.
If the NEO would have attained specified age and service levels within two years following the change in control, then the NEO is entitled to (1) subsidized and/ or unreduced pension benefits upon commencement of pension benefits in accordance with plan terms after termination of employment, and (2) subsidized retiree medical benefits as a retiree under our health plans commencing immediately after the expiration of the benefit continuation period at active-employee rates as described above.
An NEO who is a participant in the Company’s pension plan will become vested (if not already) in the applicable accrued benefit as of the termination date.
Continued financial planning benefits and outplacement assistance benefits for up to 12 months.
A “change in control” for purposes of the Change in Control Plan generally consists of any of the following:
An NEO who is a participant in the Company’s pension plan will become vested (if not already) in the applicable accrued benefit as of the termination date;
Continued financial planning benefits for up to 12 months; and
Outplacement services for a period of time as provided under the Separation Plan.
An NEO is not eligible for benefits under the Change in Control Plan following a termination due to death or permanent disability.
For purposes of the Change in Control Plan:
A “change in control” generally consists of:
| (i) | an acquisition of more than 30% of the Company’s voting securities (other than acquisitions directly from the Company); |
| (ii) |
|



















































 Allow Directors and management employees, including officers, to engage in transactions involving short sales, publicly traded options, hedging or pledging of
Allow Directors and management employees, including officers, to engage in transactions involving short sales, publicly traded options, hedging or pledging of